EED
Two fragments of human EED (amino acids 40–441 and 76–441) covering the WD40 repeats were subcloned into a modified pET28 GST-LIC vector. The recombinant proteins were overexpressed at 16 °C in Escherichia coli BL21 (DE3) Codon plus RIL (Stratagene) as N-terminal GST-tagged fusion proteins and were purified by affinity chromatographyon glutathione-sepharose (GE Healthcare).
Lysis buffer and binding buffer: 20 mM Tris, pH 7.5, 400 mM NaCl, with or without 1 mM DTT
elution buffer: 20 mM Tris, pH 7.5, 400 mM NaCl, 15 mM Glutathione
After cleavage using thrombin (Sigma Aldrich) on the column at room temperature for 4–5 h, the flow-through was collected and purified further by size exclusion chromatography(Superdex 200; GE Healthcare). gel filtration buffer: 20 mM Tris, pH 7.5, 1 mM DTT, 200 mM NaCl.
The proteins were collected and then concentrated to 15 mg∕mL in a buffer containing 20 mM Tris-HCl, pH 7.5, 0.2 M NaCl, and 1 mM DTT.
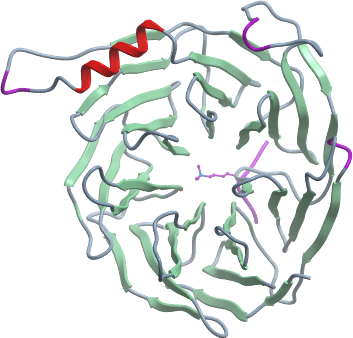
Crystallization:Before crystallizatin, Concentrated EED WD40 was mixed with H4K20me3 peptide at ration of 1:3. Crystals of human EED WD40 domain in complex with H4K20me3 was grown at 291 K using the hanging drop method by mixing equal volumes of 0.1M BTP, 3.5M sodium formate, 10 mM TCEP chloride, and 15 mg/mL protein/ligand complex. The crystals were cryoprotected by cryoprotectant consisting of 100% reservoir solution.