PTPN11, also known as SH2 containing protein tyrosine phosphatase 2 (SHP2) plays an important regulatory role in a number of diverse cell signaling events controlling mitogenic activation, metabolism, transcription regulation, cell migration, immune regulation as well as development. SHP2 is widely expressed in most tissues with particular high expression in heart, brain, and skeletal muscle.
Due to its central role in signaling deregulation of SHP2 has been linked to a large number of diseases. In contrast to most phosphatases SHP2 acts positively on signaling and is a bona fide oncogene. Mutations in SHP2 are the cause of Noonan syndrome, an autosomal dominant disorder characterized by dysmorphic facial features, proportionate short stature and heart disease (most commonly pulmonic stenosis and hypertrophic cardiomyopathy). Webbed neck, chest deformity, cryptorchidism, mental retardation and bleeding diatheses also are frequently associated with this disease. This syndrome is relatively common, with an estimated incidence of 1 in 1,000-2,500 live births; all mutations are missense mutations causing gain of function of the phosphatase and are present in 50% of Noonan syndrome individuals.
Mutations in SHP2 have also been associated with acute myeloid leukemia. Germline mutations in SHP2 have been reported in juvenile myelomonocytic leukemia (JMML) and somatic SHP2 mutations account for 34% of non-syndromic JMML. Mutations in SHP2 are also found in a small percentage of individuals with myelodysplastic syndrome (MDS) and de novo acute myeloid leukemia (AML).
In addition, mutations in SHP2 have been associated with the Leopard syndrome, resulting in multiple lentigines, electrocardiographic-conduction abnormalities, ocular hypertelorism, pulmonary stenosis, abnormal genitalia, retardation of growth, and sensorineural deafness. This syndrome is an autosomal dominant condition--characterized by lentigines and café au lait spots, facial anomalies, cardiac defects that shares several clinical features with Noonan syndrome (NS).
Furthermore, SHP2 is one of the targets of the Helicobacter pylori CagA protein which is injected from attached Helicobacter pylori into host cells in the stomach resulting in the formation of gastric carcinoma. SHP2 is also a potential therapeutic target for leptin resistance and obesity.
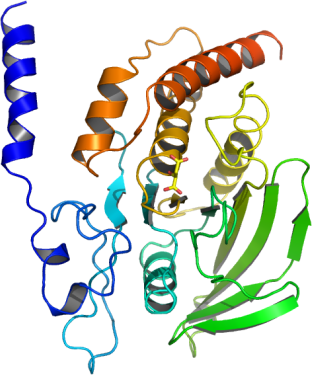
The SHP2 N-terminus represses phosphatase activity by interacting with the PTP domain blocking the catalytic site. The current crystal structure of full length SHP2 has therefore an inaccessible active site and cannot be used for cocrystallization of SHP2 with inhibitors. We therefore determined here a high resolution structure of an open conformation of SHP2.