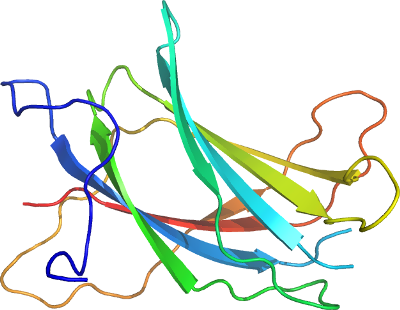
NEDD4
PDB:3B7Y
Entry Clone Accession:NP_006145
Entry Clone Source:nedd4.BAA07655.KAZUSA.KIAA0093.pBluescriptSK+
SGC Clone Accession:nedd4.0001.0152.94E03 (SDC094E03)
Tag:N-terminal: MHHHHHHSSGRENLYFQ*G (removed)
Host:BL21 (DE3)
Vector:p28a-LIC-TEV
Sequence: mhhhhhhssgrenlyfq*gMATCAVEVFGLLEDEENSRIVRVRVIAGIGLAKKDILGASDPYVRVTLYDPMNGVLTSVQTKTIKKSLNPKWNEEILFRVHPQQHRLLFEVFDENRLTRDDFLGQVDVPLYPLPTENPRLERPYTFKDFVLHPRSHKSRVKGYLRLKMTYLP
Growth
Medium:TB
Procedure: A 250 ml flask containing LB (Sigma L7658) supplemented with 50 ug/ ml kanamycin (BioShop Canada KAN 201) was inoculated from a glycerol stock of the bacteria. The flask was shaken overnight (16 hours) at 250 rpm at 37 degC.
Using the Lex system, a 2L bottle (VWR 89000-242) containing 1800 ml of TB (Sigma T0918) supplemented with 1.5% glycerol, 50 ug/ ml kanamycin and 600 ul antifoam 204 (Sigma A-8311) was inoculated with 50 ml overnight LB culture, and incubated at 37 degC. The temperature of the media was reduced to 15 degC one hour prior to induction and induced at OD(600) = 6 with 100 uM isopropyl-thio-ß-D-galactopyranoside (BioShop Canada IPT 001). Cultures were aerated overnight (16 hours) at 15 degC, and cell pellets collected by centrifugation and frozen at -80 degC.
Purification
Procedure:
IMAC: Unclarified lysate was mixed with 2-3 mL of Talon Metal Affinity Resin (Clontech Ref 635504) per 100 mL lysate. The mixture was incubated with mixing for at least 60 minutes at 4 degC. The lysate was spun at 500 xg for 5 minutes, and the supernatant was decanted. The remaining resin was washed with 45 mL of cold wash buffer, allowing 5 minutes to settle between washes, until the supernatant was clear (usually 3-5 washes). The washed resin was transferred to a gravity column and further washed with 1 column volumes (approx. 5 mL) of wash buffer at approximately 3 mL/min. Samples were eluted from the resin by exposure to 2-3 column volumes (approx. 10 mL) of elution buffer.
His-tag cleavage: An aliquot of homemade TEV was added to the IMAC eluate to approx. 10 ug TEV / mg protein and incubated overnight at 4 degC prior to gel filtration.
Gel filtration chromatography: An XK 16x65 column (part numbers 18-1031-47 and 18-6488-01, GE Healthcare) packed with HighLoad Superdex 200 resin (10-1043-04, GE Healthcare) was pre-equilibrated with gel filtration buffer for 1.5 column volumes using an AKTAxpress (18-6645-05, GE Healthcare) at a flow rate of 1.5 mL/min. The eluate sample from the IMAC step (approx. 10 mL) was loaded onto the column at 1.5 mL/min, and 1mL fractions were collected into 96-well plates (VWR 40002-012) using peak fractionation protocols). Fractions observed by a UV absorption chromatogram to contain the protein were pooled.
Protein yield was 1 mg per liter of bacterial culture.
Extraction
Procedure: Frozen cell pellet contained in bags (Beckman 369256) obtained from 4L of culture were thawed by soaking in warm water. Each cell pellet was resuspended in 25-40 mL lysis buffer and homogenized using an Ultra-Turrax T8 homogenizer (IKA Works) at maximal setting for 30-60 seconds per pellet. Cell lysis was accomplished by sonication (Virtis408912, Viewsonic) on ice: the sonication protocol was 10 sec pulse at half-maximal frequency (5.0), 10 second rest, for 10 minutes total sonication time per pellet.
Concentration:Protein concentration. Purified protein was concentrated using 15 mL concentrators with a 5,000 molecular weight cut-off (Amicon Ultra-15, UFC900524, Millipore) at 3750 rpm, typically resulting in a final concentration of 7-10 mg/mL.
Structure Determination
MassSpec:Mass-spectroscopy by LCMS shows that the product was pure and of correct molecular weight.
Crystallization:Crystals of the Nedd4 C2-like domain were grown at 298 K using the hanging drop method by mixing 2 volumes of 2 mg/ml protein with 1 volume of solution of 10% PEG4000, 0.1 Succinic acid and adding Spermine tetra-HCl directly into the premixed drop to a concentration of 10mM (original additive D3 from Hampton Research Additive Screen HT-HR2-138). The crystals were cryoprotected by gradual substitution of crystallization buffer in the drop to 30% PEG4000.