MERTK
PDB:3BPR
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:NP_003749.2
Entry Clone Source:axl2.LIFESEQ1708542.OBS.IMAGE:1708542.pINCY
SGC Clone Accession:SDC037B11
Tag:N-terminal: MGSSHHHHHHSSGLVPRGS
Host:BL21 (DE3)
Construct
Prelude:
Sequence:mgsshhhhhhssglvprgsEELQNKLEDVVIDRNLLILGKILGEGEFGSVMEGNLKQEDGTSLKVAVKTMKLDNSSQREIEEFLSEAACMKDFSHPNVIRLLGVCIEMSSQGIPKPMVILPFMKYGDLHTYLLYSRLETGPKHIPLQTLLKFMVDIALGMEYLSNRNFLHRDLAARNCMLRDDMTVCVADFGLSKKIYSGDYYRQGRIAKMPVKWIAIESLADRVYTSKSDVWAFGVTMWEIATRGMTPYPGVQNHEMYDYLLHGHRLKQPEDCLDELYEIMYSCWRTDPLDRPTFSVLRLQLEKLLESLPDV
Vector:pET28a-LIC
Growth
Medium:TB
Antibiotics:
Procedure:A small overnight culture containing 50 µg/mL kanamycin was used to inoculate TB media containing the same concentration of antibiotics. Cultures were grown at 37°C for about 6 hours until the OD600 reached ~0.8, the temperature was adjusted to 15°C, and expression was induced using 0.1mM IPTG overnight. Cells were harvested by centrifugation and frozen.
Purification
Procedure
Clarified supernatant was mixed with 5.0 ml 50% Talon resin slurry (Clonetech), rotated for 1 hour at 4 degrees Celsius, and then loaded into a column. Ten column volumes of washing buffer were used for washing before elution with 7 mL elution buffer.
Gel-filtration was conducted using AKTAxpress (18-6645-05, GE Healthcare) with XK 16x65 columns (part numbers 18-1031-47 and 18-6488-01, GE Healthcare) packed with HighLoad Superdex 200 resin (10-1043-04, GE Healthcare). Pre-equilibration was done with gel filtration buffer at a flow rate of 3 mL/min. Seven mL of protein sample was loaded onto the column at 1.5 mL/min, and 2mL fractions are collected in 96-well plates (VWR 40002-012) using peak fractionation protocols with the following parameters: (Slope; min. peak width 0.833 min; level 0.000 mAU; peak start slope 10.000 AU/min; peak end slope 20.000 AU/min). Fractions were analyzed for purity using SDS-PAGE and those containing pure Mer/axl2 were pooled.
Extraction
Procedure
Frozen cell pellets obtained from 2L culture were thawed, resuspended in 50 mL lysis buffer, homogenized with Ultra-Turrax T8 homogenizer (IKA Works) at maximal setting for 30-60 seconds, and twice passed through a microfluidizer (Microfluidics M110EH) at 18,000 psi. The lysate was clarified by centrifugation (JA25.50 rotor, Avanti J-20 XPI, Beckman Coulter) for 20 minutes at 69,673 x g.
Concentration:Purified protein was concentrated using 15 mL concentrators with a 10,000 molecular weight cut-off (Amicon Ultra-15, UFC900524, Millipore) at 3750 rpm, typically resulting in a final concentration of 7-10 mg/mL.
Ligand
MassSpec:Mass-spectroscopy by LCMS shows that the product was pure and of correct molecular weight.
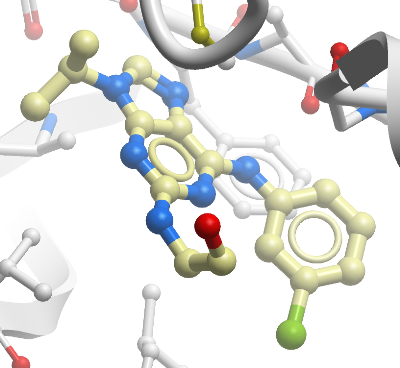
Crystallization:2-(2-hydroxyethylamino)-6-(3-chloroanilino)-9isopropylpurine (Calbiochem - 50 mM in DMSO) was added to 8 mg/ml Mer protein to the final concentration of 2.5 mM. The mixture was rocked at 4 degC overnight and further concentrated to about 35 mg/ml.Crystals were grown by mixing 2 microL Mer inhibitor solution and 2 microL reservoir solution (100 mM Tris-HCl pH 8.5, 3.64 M NaCl) at 14 degC using the hanging-drop vapour-diffusion method. Crystals were cryo-protected with a solution composed of glycerol, ethylene glycol, glucose, and fructose.
NMR Spectroscopy:
Data Collection:
Data Processing: