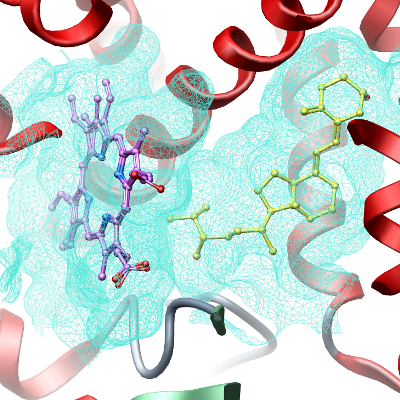
Cytochrome P450 (CYP) superfamily of enzymes are ubiquitous heme-containing monooxygenases named for the absorption band at 450 ηm of their reduced carbon monoxide (CO) form. The CYP enzymes are responsible for the metabolism of xenobiotics, including drugs, carcinogens, and environmental chemicals, as well as endogenous compounds such as steroids and fatty acids, thereby participating in the maintenance of the body homeostasis. CYP2R1 belongs to the CYP2 family of proteins, which are frequently involved in the metabolism of foreign compounds. CYPR1 gene deficiency cause inherited rickets due to a defect in vitamin D 25-hydroxylation. CYP2R1 catalyzes the initial step of activation of vitamin D3 (cholecalciferol) to its metabolite 25-hydroxycholecalciferol (the precursor of biologically active 1,25-dihydroxycholecalciferol). CYP2R1 has been shown to be active in the 25-hydroxylation of vitamin D2 (Cheng et al., PNAS 2003), but no xenobiotic substrate for this enzyme has been identified so far. CYP2R1 is expressed in liver and highly conserved across species from Fugu to Human. In contrast, most other members of the CYP2 family show a high degree of divergence, with little conservation.
Here we present the crystal structure of human CYP2R1 in complex with vitamin D2.