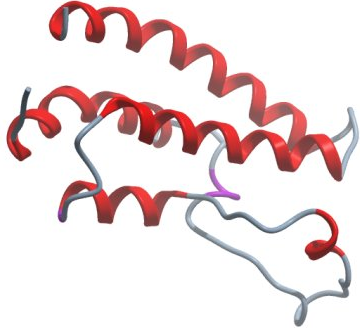
|
Entry Clone Source: Synthetic
|
|
Entry Clone Accession: n/a
|
|
SGC Construct ID: CREBBPA-c003
|
|
GenBank GI number: gi|4758056
|
|
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ]
|
|
Tags and additions: Tag sequence: mhhhhhhssgvdlgtenlyfq*sm, cleaved at the * with TEV protease.
|
|
Expressed sequence: (tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfq*smRKKIF
KPEELRQALMPTLEALYRQDPESLPFRQP
VDPQLLGIPDYFDIVKNPMDLSTIKRKLD
TGQYQEPWQYVDDVWLMFNNAWLYNRKTS
RVYKFCSKLAEVFEQEIDPVMQSLG
|
|
Host: BL21(DE3)-R3: a phage resistant BL21(DE3) derivative
|
|
Growth medium, induction protocol
Host cells transformed with the expression plasmids were plated out onto LB-agar plates containing 50 mg/ml kanamycin. The next day several colonies were combined into 1 ml TB (Terrific Broth), 50 µg/ml kanamycin, which was then grown overnight and stored as glycerol stocks at -80°C.
The glycerol stock was used to inoculate a 10-ml starter culture in TB + kanamycin (50 µg/ml). This starter culture was grown overnight at 37°C and used to inoculate a 1 liter culture in the same medium. The culture was grown in baffled flasks at 37°C until the OD600reached ~3.5. After that the temperature was lowered to 18°C. Protein production was induced with 0.1mM IPTG and the recombinant bromodomain was incubation continued at 18°C overnight. The next day cells were harvested by centrifugation at 4000 rpm for 30 minutes. The cell pellet was stored at -80°C degrees
|
|
Extraction buffer and extraction method: Lysis buffer: 500mM NaCl, 50mM pH8.0 KH2PO4, 0.5mM TCEP, Benzonase 1µl/15 ml buffer, Protease inhibitor (1 µl/ml); Afinity binding buffer: 10mM Imidazole, 500mM NaCl, 50mM pH8.0 KH2PO4, 0.5mM TCEP.
|
| Procedure: The cell pellet (40g) from 4 L culture was re-suspended in one volume (40 ml) of lysis buffer. The re-suspended cells were lysed by one passage through a Constant Systems cell breaker and subsequent sonication; the cell breaker was washed with 1x extraction buffer, bringing the total volume to 120 ml. DNA was precipitation by addition of polyethyleneimine (PEI, pH 7.5) to a final concentration of 0.15 % during an incubation time of 30 min on ice, followed by a centrifugation at 17,000 rpm (4°C); The supernatant was further cleared by filtration through a 0.2 µm serum Acrodisc filter. |
|
Column 1: Ni-affinity chromatography: HisTrap FF Crude, 5 ml (GE Healthcare).
|
| Buffers: Binding Buffer: 50mM NaH2PO4, 500mM NaCl, 30mM Imidazole, pH 8.0, 0.5mM TCEP; Elution Buffer: 50mM NaH2PO4, 500mM NaCl, 250mM Imidazole, pH 8.0, 0.5mM TCEP. |
|
Procedure: All purification steps were carried out using an AKTAexpress system (GE Healthcare) at 7ºC. The lysate was loaded on a pre-equilibrated His-trap column at 0.8 ml/min. After loading, the column was washed at 0.8 ml/min with 50 ml binding buffer and the protein was eluted with 25 ml of elution buffer. The peak fraction was collected automatically according to A280.
|
|
Enzymatic treatment and Tag removal: TEV protease (1:20 w/w), was added to the sample after gel filtration. The sample was incubated at 4°C overnight. The sample was then passed over a column of Ni-sepharose (0.5 ml) to trap the cleaved tag and other Ni-binding proteins.
|
|
Column 2: Size exclusion chromatography HiLoad 16/60 Superdex 75
|
| SEC-Buffers: 10mM Hepes, pH 7.4, 500 mM NaCl, 5% glycerol, 0.5mM TCEP |
| Procedure: Fractions containing the expressed bromo domain were collected after his6-tag cleavage and were loaded on a SEC column at 1.0 ml/min. Eluted fractions were >95% pure as judged by SDS-PAGE |
|
Mass spec characterization (After tag cleavage): Expected MW: 14207 (cleaved protein); Measured MW: 14207
|
|
Protein concentration: The protein was concentrated to 10 mg/ml in SEC buffer using a centricon device with a 10kDa cut off
|
|
Crystallization: The protein (10 mg/ml) in SEC buffer was mixed with an equal volume (100 nl) of reservoir solution (0.2M Potassium thiocyanate; 25% (w/v) PEG3350; 5% (v/v) ethylene glycol and equilibrated as a sitting drop at 4°C.
|
| Data Collection: Crystals were flash frozen in liquid nitrogen using the crystallization condition supplemented with 25% ethylene glycol. Diffraction data were collected to 1.98 Å at a RIGAKU FR-E+ SuperBright light source at a single wavelength of 1.54128 Å. |