Calcium dependent protein kinases (CDPK) are found only in some plants - e.g. Arabidopsis thanalia - and some protozoan organisms, including Plasmodium, Cryptosporidium and Toxoplasma parasites.
We have previously determined the apo structure of kinase domain of
(encoded by the gene cgd7_1840.
From Stargazer screening, we identified a number of kinase inhibitors which stabilized CpCDPK8, including staurosporine and indirubin derivative E804.
In addition, we have determined determined that indirubin E804 effectively inhibited the enzyme's phosphorylation activity with a IC50 value of 1.2 μM.
Indirubins (IRD) are a class of natural chemicals extracted from the indigo plant as well as gastropods of the Muricidae and Thaididae families (Polychronopoulos et al., 2004). They are also the active ingredients in the Chinese herbal mixture Dang Gui Long Hui Wan, which was found to have antileukemic activity back in 1980's. The mechanism of action is based on inhibition of both S/T and tyrosine protein kinases including cyclin-dependent kinases (CDK), glycogen synthase kinase (GSK) and Stat3 (Meijer et al., 2003; Polychronopoulos et al., 2004).
We were able to crystallize CpCDPK8's kinase domain with indirubin 804 bound - the first ever complex of a CDPK with a ligand. As noted in most kinases, the presence of a ligand in the nucleotide cleft bends the N lobe towards the C-lobe, particularly the flexible glycine-rich region.
The backbone interaction between the hinge and ligand is conserved in known kinases/indirubin complexes, as shown in the table below.
Different indirubin derivatives are distinguished by different moieties at the 3'and 5-positions on the bisindole.
Structure
Ligands
Interacting residues
CpCDPK8 (3F3Z)
Indirubin E804
Glu103
Cys105
hCaMK2A (2VZ6)
Indirubin E804
Asp90
Val92
hCDK5 (1UNH)
Indirubin 3'-monoxime
Glu81
Cys83
hCDK2 (1E9H)
Indirubin 3'-monoxime-5-sulphonic acid
Glu81
Leu83
hGSK3 (1Q41)
Indirubin 3'-monoxime
Asp133
Val135
CpGSK (3EB0)
Indirubin E804
Glu118
Val119
Indirubin 804 is distinguished by its side chain on the bisindole's 3'position.
This is an important moiety because other indirubin compounds in our screening library, all with the same bisindole structure but different decorations at the 3'or 5 positions, failed to stabilize CpCPDK8.
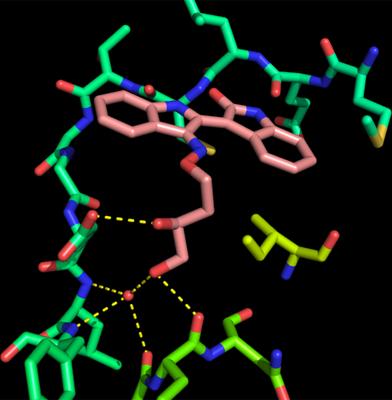
The figure above shows how this side chain forms polar interaction with Glu109, Glu152 and, via a water molecule, Leu110, Phe111 and Pro151.
These hydrogen bonds result partly from the polar side chain being forced by the hydrophobic side chain of Ile168 (located just before the DFG triad) to bend in a direction perpendicular to the plane of the bisindole structure.
In the absence of the hydrophobic residue, the IRD molecule would adopt the conformation found in the hCaMK2A structure.
The presence of this nonpolar residue and the resulting change in the confomation of IRD804 are unique to CpCDPK8 and may be potentially exploited for development of a selective inhibitor.
Based on our biochemical assays, indirubin compounds are potentially promising anticryptosporidial drug candidates. In particular, IRD804 is able to inhibit CpCPDK8 at low micromolar concentrations. Our structural complex of CpCDPK8 and IRD804 provides evidence of a unique mechanism of inhibition that can be used to develop a more selective and more potent inhibitor. Certainly, it gives us motivation to test other indirubin derivatives with similar side chain decoration.