Entry Clone Source: Ordered-synthetic |
Entry Clone Accession: n/a |
SGC Construct ID: CA6A-c110 |
GenBank GI number: gi|70167127 |
Vector: pNIC-CTHF. Details [ PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Tags and additions: C-terminal, TEV cleavable hexahistidine tag. Tag sequence: enlyfq(*)shhhhhhdykddddk |
Host:E. coli BL21(DE3)-R3 |
Sequence (tag sequence in lowercase):
MSDWTYSEGALDEAHWPQHYPACGGQRQS
PINLQRTKVRYNPSLKGLNMTGYETQAGE
FPMVNNGHTVQISLPSTMRMTVADGTVYI
AQQMHFHWGGASSEISGSEHTVDGIRHVI
EIHIVHYNSKYKSYDIAQDAPDGLAVLAA
FVEVKNYPENTYYSNFISHLANIKYPGQR
TTLTGLDVQDMLPRNLQHYYTYHGSLTTP
PCTENVHWFVLADFVKLSRTQVWKLENSL
LDHRNKTIHNDYRRTQPLNHRVVESNFPN
QEYTLGSEFQAenlyfq
enlyfq residues originate from the vector and remain after the TEV cleavage of the hexahistidine tag. |
Expression: 10 µl of BL21(DE3)-R3 glycerol stock were inoculated into 100ml of TB with 50µg/ml of kanamycin and 34µg/ml chloramphenicol and grown overnight at 37°C, 200rpm. 10ml of overnight culture were added to 1L of TB with 50µg/ml kanamycin and incubated at 37°C, 160rpm. After the OD600 reached 1.0, the temperature was dropped to 18°C and 500µl of 1M IPTG was added to the final concentration of ~0.5mM. The culture was then incubated with shaking overnight at 18°C, 160rpm. The following morning the 4L culture was harvested and centrifuged for 10min at 4000rpm. Supernatant was discarded and cell pellets were resuspended in 80ml of a lysis buffer and frozen at -80°C. |
Extraction: Lysis buffer: 50mM HEPES pH 7.5, 500mM NaCl, 5mM Imidazole, 5% glycerol + EDTA-free Complete (1 tablet/50ml). The thawed cells were broken by 5 passes at 16.000 psi through a high pressure homogeniser followed by centrifugation for 45 min at 15.000rpm. |
Purification:
Column 1: Ni-affinity, His-Trap, 1 ml (Amersham)
Column 2: Superdex 200, HiPrep 16/60 (Amersham) |
Buffers: Start buffer: 50mM HEPES pH 7.5, 500mM NaCl, 20mM Imidazole, 5% glycerol, EDTA-free Complete, 0.5mM TCEP; Washing buffer: 50mM HEPES pH 7.5, 500mM NaCl, 40mM Imidazole, 5% glycerol, EDTA-free Complete, 0.5mM TCEP; Elution buffer: 50mM HEPES pH 7.5, 500mM NaCl, 5% glycerol, 250mM Imidazole, 0.5mM TCEP; GF buffer: 10mM HEPES pH 7.5, 150mM NaCl, 5% glycerol, 0.5mM TCEP. |
Procedure: The cell extract was loaded on the AKTA Express system, extinction at 280nm was monitored and fractions were collected and analyzed by SDS-PAGE. Positive fractions were pooled for TEV cleavage. |
TEV cleavage: The His-tag was cleaved with 1mg TEV per 40 mg target protein at 4°C overnight. The protein was purified on IMAC Sepharose using buffers as above. Protein was characterised by ESI-TOF mass spectrometry. |
Concentration and buffer exchange: Using Amicon Ultra-15 concentrators with 10 kDa cutoff, the sample was buffer-exchanged into GF buffer and concentrated to 9mg/ml. Concentrations were determined from the absorbance at 280 nm using a NanoDrop spectrophotometer. |
Mass spectrometry characterization: Calculated mass of the construct was 31914. Experimental mass determination indicates loss of the N-terminal Met residue, however a yet undefined loss of 35 Da is observed. |
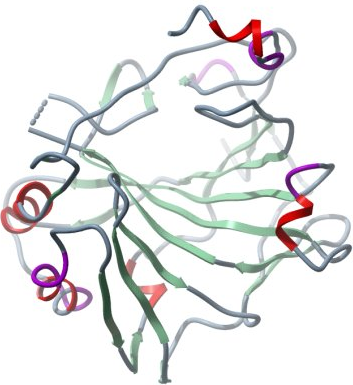
Crystallisation: Crystals were grown by vapor diffusion at 4°C in 150nl sitting drops. The drops were prepared by mixing 50nl of protein solution and 100nl of precipitant consisting of 0.16M MgCl2, 0.08M Tris pH 8.5, 16% w/v PEG3350. Crystals were transferred to a cryo-protectant consisting of 25% ethylene glycol and 75% well solution before flash-cooling in liquid nitrogen. |
Data Collection: Resolution: 1.9Å; X-ray source: SLS beam X10SA. |