Entry Clone Source: Synthetic |
Entry Clone Accession: n/a |
SGC Construct ID: TAF1LA-c061 |
GenBank GI number: gi|24429572 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
CATATGCACCATCATCATCATCATTCTTC
TGGTGTAGATCTGGGTACCGAGAACCTGT
ACTTCCAATCCATGCAGGTGGCCTTTAGC
TTTATCCTGGATAATATCGTGACCCAGAA
AATGATGGCGGTGCCGGATAGCTGGCCGT
TTCATCATCCGGTTAACAAAAAATTTGTT
CCGGATTATTATAAAATGATTGTGAATCC
GGTTGATCTGGAAACCATCCGTAAAAATA
TTAGCAAACATAAATATCAGAGCCGCGAA
AGCTTTCTGGATGATGTGAACCTGATTCT
GGCCAACAGCGTTAAATATAACGGTCCGG
AAAGCCAGTATACCAAAACCGCGCAGGAA
ATTGTGAATATTTGCTATCAGACCATTAC
CGAATATGATGAACATCTGACCCAGCTGG
AAAAAGATATTTGCACCGCGAAAGAAGCG
GCGCTGGAAGAAGCCGAACTGGAAAGCCT
GGATTGACAGTAAAGGTGGATACGGATCC
GAA |
Tags and additions: Cleavable N-terminal His6 tag. |
Final protein sequence:
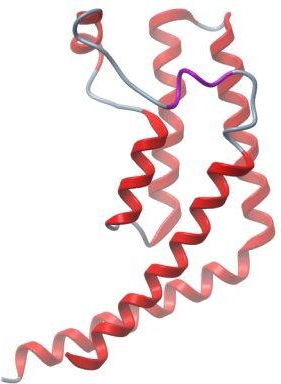
mhhhhhhssgvdlgtenlyfq^sMQVAFS
FILDNIVTQKMMAVPDSWPFHHPVNKKFV
PDYYKMIVNPVDLETIRKNISKHKYQSRE
SFLDDVNLILANSVKYNGPESQYTKTAQE
IVNICYQTITEYDEHLTQLEKDICTAKEA
ALEEAELESLD
^ TEV cleave site |
Host: BL21 (DE3)R3-pRARE2 (Phage resistant strain) |
Growth medium, induction protocol: 10 ml from a 50 ml overnight culture containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol were used to inoculate each of two 1 litre cultures of TB containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol. Cultures were grown at 37°C until the OD600 reached ~2.5then the temperature was adjusted to 18°C. Expression was induced overnight using 0.1 mM IPTG at an OD600 of 3.0. The cells were collected by centrifugation and the pellet resuspended in binding buffer and frozen. Binding buffer: 50 mM HEPES pH 7.5; 500 mM NaCl; 10 mM imidazole, 5% glycerol. |
Extraction buffer, extraction method: Frozen pellets were thawed and fresh 0.5 mM TCEP, 1 mM PMSF added to the lysate. Cells were lysed using sonication. The lysate was centrifuged at 16,500 rpm for 60 minutes and the supernatant collected for purification. |
Column 1: Ni-affinity. Ni-sepharose (Amersham), 5 ml of 50% slurry in 1.5 x 10 cm column, washed with binding buffer. |
Buffers : Binding buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 5 mM imidazole;
Wash buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 30 mM Imidazole;
Elution buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 50 to 250 mM Imidazole, (step elution). |
Procedure: Supernatant was loaded by gravity flow on the Ni-sepharose column. The column was then washed with 30 ml wash buffer at gravity flow. The protein was eluted by gravity flow by applying 5-ml portions of elution buffer with increasing concentration of imidazole (50 mM, 100 mM, 150 and 250 mM); fractions were collected until essentially all protein was eluted. |
Column 2: Size Exclusion Chromatography. Superdex S75 16/60 HiLoad |
Buffers: 10 mM HEPES, pH 7.5; 500 mM NaCl, 5% glycerol |
Procedure: TAF1L was concentrated and applied to an S75 16/60 HiLoad gel filtration column equilibrated in 10 mM HEPES, pH 7.5; 500mM NaCl, 5% glycerol using an ÄKTAexpress system. |
Mass spectrometry characterization: LC- ESI -MS TOF gave a measured mass of 18000 for this construct as predicted from the sequence of this protein. |
Protein concentration: Protein was concentrated to 10.8 mg/ml using an Amicon 3 kDa cut-off concentrator. |
Crystallization: Crystals were grown at 4°C in 300 nl sitting drops from a 1:2 ratio of protein to reservoir solution containing 15 % PEG3350, 0.17 M (NH4)2SO4, 15 % glycerol. |
Data Collection: Crystals were cryo-protected using the well solution supplemented by 20 % ethylene glycole and flash frozen in liquid nitrogen; X-ray source: Diffraction data were collected from a single crystal on a Rigaku FR-E SuperBright at a single wavelength of 1.5 Å and the structure was refined to 2.06 Å;
Phasing: The structure was solved by molecular replacement using an ensemble of known bromodomain structures as a starting model. |