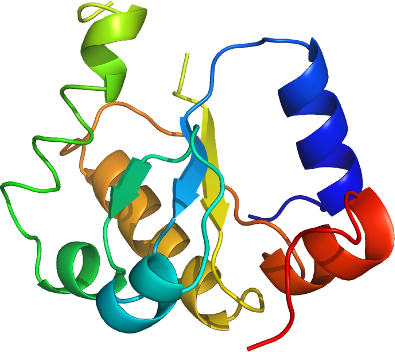
MOCS3
PDB: 3I2V
Entry Clone Accession: BC015939
Entry Clone Source: uba10.BC015939.MGC.AT11-D2.pCMV-SPORT6
SGC Clone Accession: uba10.335.460.046D11 (SDC046D11)
Tag: N-terminal tag: MGSSHHHHHHSSGLVPRGS
Host: BL21 (DE3)
Vector: pET28a-LIC
Sequence (with tag):
mgsshhhhhhssglvprgsSRVSVTDYKRLLDSGAFHLLLDVRPQVEVDICRLPHALHIPLKHLERRDAESLKLLKEAIWEEKQGTQEGAAVPIYVICKLGNDSQKAVKILQSLSAAQELDPLTVRDVVGGLMAWAAKIDGTFPQY
Growth
Procedure: Competent BL21 (DE3) cells (Invitrogen, C6000-03) were transformed and grown using the LEX system (Harbinger, BEC) at 37 °C in 2L bottles (VWR, 89000-242) containing 1800 ml of TB (Sigma, T0918) supplemented with 150 mM glycerol, 100 µM Kanamycin and 600 µl antifoam 204 (Sigma A-8311). When the OD600 reached a value of about 6.0, the temperature was reduced to 15 °C, and one hour later the culture was induced with 100 µM IPTG (BioShop, IPT001) and incubated overnight (16 hours) at 15 °C. Cell pellets were collected by centrifugation (12,227 xg, 20 mins), frozen in liquid nitrogen, and stored at -80 °C. To produce the SeMet derivative, the same bacterial cells were grown using M9 SeMet High-Yield growth media kit package (Medicilon, cat#) according to manufacturer's instructions.
Protein Purification
Procedure: Beads were transferred into a 25 mL Econo-Column (Bio-Rad 732-1010) and washed with 3 x 15 mL Wash buffer. Protein was eluted with 10 mL of Elution Buffer. Samples were gel-filtered (XK 16x65 packed with HighLoad Superdex 200 resin, GE Healthcare) using an AKTAxpress (GE Healthcare, 18-6645-05) at a flow rate of 1.5 mL/min Gel-filtration buffer, and 3 mL fractions were collected in 5 ml tubes. Fractions containing protein were pooled and concentrated using a 3 KDa MW cut-off concentrator (Millipore, UFC900524) at 2851 xg to a final value of 10 mg/mL. Protein yield was 16 mg per liter of bacterial culture. Coomassie-stained SDS-PAGE showed that the product was pure (Figure 2, inset (jp, please prepare thanks, see 9pdb_prototype) and mass-spectroscopy by LCMS (Agilent 1100 Series) showed that the protein had the correct molecular weight.
Extraction
Procedure: After resuspension in 30 mL Lysis buffer per liter bacterial culture, cells were lysed by sonication ice (Virtis408912, Virsonic: 10 sec, 50 % power, 10 sec rest, 10 min total sonication time per pellet). A volume of 2.0 mL settled TALON metal-affinity resin (BD Bioscience) per 40 mL lysate (Promega, V882A) was rocked with unclarified lysate for 60 minutes at 4 °C.
Concentration:10 mg/ml
Structure Determination
Crystallization: Crystals were grown at 18 degC using the hanging drop method by mixing equal volumes of protein (10 mg/ml) and Crystallization Buffer (21% PEG3350, 0.1 M HEPES pH 7.5). Suitable crystals were cryoprotected by immersion in well solution supplemented with cryoprotectant solution (9% sucrose (wt/vol), 2% glucose (wt/vol), 8% glycerol (vol/vol), 8% ethylene glycol (vol/vol)) prior to dunking and storage in liquid nitrogen.
Data Processing: Diffraction data from 2 crystals (1.5 and 1.25 Å highest resolution, collected at 0.964 and 1.072 Å λ, respectively) of the MOCS-RLD were collected at beamline 23-ID at the Advanced Photon Source (APS). All data sets were integrated and scaled using the HKL2000 program packages. The structure was solved by SAD phasing techniques using the software package PHENIX and an initial model was built using arp/warp with the 1.5 Å resolution data set. This initial model was then used for further model building and refinement using the 1.25 Å resolution data set. Iterative model building using the graphics program Coot, and the program REFMAC5 were used for initial rounds of refinement. Final rounds of refinement were performed using phenix.refine, that led to a final model with an R factor of 16.1% (Rfree 20.4%) for data between 1.25-24.3 Å.