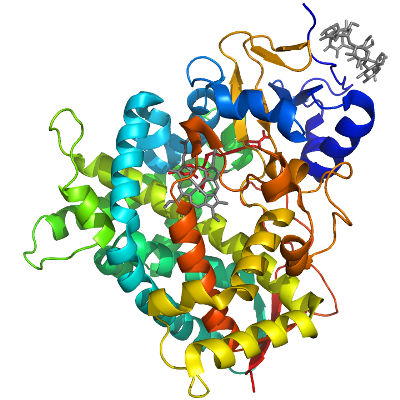
CYP51A1
PDB:3I3K
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:AT52-A8 (BC032322)
Entry Clone Source:MGC
SGC Clone Accession:
Tag:N-terminal: MAKKT;
C-terminal: 6His-tag
Host:E. coli JM109 (Stratagene).
Construct
Prelude:
Sequence:LPAGVKSPPYIFSPIPFLGHAIAFGKSPIEFLENAYEKYGPVFSFTMVGKTFTYLLGSDAAALLFNSKNEDLNAEDVYSRLTTPVFGKGVAYDVPNPVFLEQKKMLKSGLNIAHFKQHVSIIEKETKEYFESWGESGEKNVFEALSELIILTASHCLHGKEIRSQLNEKVAQLYADLDGGFSHAAWLLPGWLPLPSFRRRDRAHREIKDIFYKAIQKRRQSQEKIDDILQTLLDATYKDGRPLTDDEVAGMLIGLLLAGQHTSSTTSAWMGFFLARDKTLQKKCYLEQKTVCGENLPPLTYDQLKDLNLLDRCIKETLRLRPPVMIMMRMARTPQTVAGYTIPPGHQVCVSPTVNQRLKDSWVERLDFNPDRYLQDNPASGEKFAYVPFGAGRHRCIGENFAYVQIKTIWSTMLRLYEFDLIDGYFPTVNYTTMIHTPENPVIRYKRR
Vector:pCW-LIC-29
Growth
Medium:
Antibiotics:
Procedure:CYP51A1 was co-expressed with GroEL/ES in E. coli JM109 in TB medium. Cells were grown at 37 degC to an OD600 of 1.0 and induced by 0.5mM IPTG and 4mg/ml of arabinose and in the presence of 0.5mM δ-aminolevulinic acid and incubated 48 hours at 26°C.
Purification
Procedure
The lysate was centrifuged at 60.000g for 60min. The supernatant was loaded onto 5ml NiHiTrap column (Amersham Biosciences) equilibrated with buffer A. The column was washed with buffer A and protein was eluted using a linear gradient of 5-100% Buffer B. The protein was further purified by ion-exchange chromatography on SourceS column (Amersham Biosciences), equilibrated with buffer 5mM KPi, pH 7.4, 20% glycerol and eluted with linear gradient of Buffer C.
Extraction
Procedure
Collected/resuspended cells (50mM potassium phosphate, 300mM NaCl, 20% glycerol, pH 7.4, 0.4mM PMSF) were disrupted in a high-pressure Microfluidizer (Microfluidics Corp.) at 18.000 psi.
Concentration:20 mg/ml
Ligand
MassSpec:Expected MW is 52550, measured mass is 52551.
Crystallization:Purified CYP51A1 was crystallized in presence of ketoconazole using hanging drop vapor diffusion method drop at 18 °C by mixing 1µl of the protein solution with 1µl of the reservoir solution containing 0.1 M Hepes pH 7.5, 2.5 M Ammonium Sulfate.
NMR Spectroscopy:
Data Collection:
Data Processing: