Entry Clone Source: MGC |
Entry Clone Accession: IMAGE:3629376 |
SGC Construct ID: ADAA-c103 |
GenBank GI number: gi|47078295 |
Vector: pNIC-CTHF. Details [ PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
CTTAAGAAGGAGATATACTATGCCCGCCT
TCGACAAGCCCAAAGTAGAACTGCATGTC
CACCTAGACGGATCCATCAAGCCTGAAAC
CATCTTATACTATGGCAGGAGGAGAGGGA
TCGCCCTCCCAGCTAACACAGCAGAGGGG
CTGCTGAACGTCATTGGCATGGACAAGCC
GCTCACCCTTCCAGACTTCCTGGCCAAGT
TTGACTACTACATGCCTGCTATCGCGGGC
TGCCGGGAGGCTATCAAAAGGATCGCCTA
TGAGTTTGTAGAGATGAAGGCCAAAGAGG
GCGTGGTGTATGTGGAGGTGCGGTACAGT
CCGCACCTGCTGGCCAACTCCAAAGTGGA
GCCAATCCCCTGGAACCAGGCTGAAGGGG
ACCTCACCCCAGACGAGGTGGTGGCCCTA
GTGGGCCAGGGCCTGCAGGAGGGGGAGCG
AGACTTCGGGGTCAAGGCCCGGTCCATCC
TGTGCTGCATGCGCCACCAGCCCAACTGG
TCCCCCAAGGTGGTGGAGCTGTGTAAGAA
GTACCAGCAGCAGACCGTGGTAGCCATTG
ACCTGGCTGGAGATGAGACCATCCCAGGA
AGCAGCCTCTTGCCTGGACATGTCCAGGC
CTACCAGGAGGCTGTGAAGAGCGGCATTC
ACCGTACTGTCCACGCCGGGGAGGTGGGC
TCGGCCGAAGTAGTAAAAGAGGCTGTGGA
CATACTCAAGACAGAGCGGCTGGGACACG
GCTACCACACCCTGGAAGACCAGGCCCTT
TATAACAGGCTGCGGCAGGAAAACATGCA
CTTCGAGATCTGCCCCTGGTCCAGCTACC
TCACTGGTGCCTGGAAGCCGGACACGGAG
CATGCAGTCATTCGGCTCAAAAATGACCA
GGCTAACTACTCGCTCAACACAGATGACC
CGCTCATCTTCAAGTCCACCCTGGACACT
GATTACCAGATGACCAAACGGGACATGGG
CTTTACTGAAGAGGAGTTTAAAAGGCTGA
ACATCAATGCGGCCAAATCTAGTTTCCTC
CCAGAAGATGAAAAGAGGGAGCTTCTCGA
CCTGCTCTATAAAGCCTATGGGATGCCAC
CTTCAGCCTCTGCAGGGCAGAACCTCGCA
GAGAACCTCTACTTCCAATCGCACCATCA
TCACCACCATGATTACAAGGATGACGACG
ATAAGTGAGGATCC |
Tags and additions: C-terminal His6 tag and FLAG tag, preceded by a TEV protease cleavage site:
aenlyfq*shhhhhhdykddddk (* - TEV cleavage site) |
Expressed sequence (tag sequence in lowercase):
MPAFDKPKVELHVHLDGSIKPETILYYGR
MPAFDKPKVELHVHLDGSIKPETILYYGR
RRGIALPANTAEGLLNVIGMDKPLTLPDF
LAKFDYYMPAIAGCREAIKRIAYEFVEMK
AKEGVVYVEVRYSPHLLANSKVEPIPWNQ
AEGDLTPDEVVALVGQGLQEGERDFGVKA
RSILCCMRHQPNWSPKVVELCKKYQQQTV
VAIDLAGDETIPGSSLLPGHVQAYQEAVK
SGIHRTVHAGEVGSAEVVKEAVDILKTER
LGHGYHTLEDQALYNRLRQENMHFEICPW
SSYLTGAWKPDTEHAVIRLKNDQANYSLN
TDDPLIFKSTLDTDYQMTKRDMGFTEEEF
KRLNINAAKSSFLPEDEKRELLDLLYKAY
GMPPSASAGQNLaenlyfqshhhhhhdyk
ddddk |
Tag removed: yes |
| Host: BL21(DE3)-R3-pRARE2 |
Expression protocol: A glycerol stock of host strain BL21(DE3)-R3-pRARE2 carrying the expression plasmid was used to inoculate 10 ml of TB (terrific Broth) supplemented with 50 µg/ml kanamycin and 35 µg/ml chloramphenicol. This starter culture was grown overnight at 37°C and used to inoculate a 1 liter culture in TB supplemented with 50 µg/ml kanamycin only. The culture was grown at 37°C until the OD600 reached ˜3.0. After that the temperature was lowered to 18°C. Protein production was induced with 0.1 mM IPTG and recombinant ADAA was expressed at that temperature overnight. The next day cells were harvested by centrifugation at 5000 rpm for 20 minutes and pellets were frozen at -80°C. |
Cell extraction:
2x Lysis buffer: 100 mM HEPES, pH 7.5, 1 M NaCl, 20 mM imidazole, 10% glycerol, 1 mM TCEP, 2x Protease Inhibitors Cocktail Set VII (Calbiochem, 1/1000 dilution), Benzonase (3 µl per 25 ml, 30 units/ml).
Procedure:
Frozen cell pellets were thawed at room temperature for approximately 1 hour then transferred to ice. One volume (i.e. 1 ml for every gram of cells) of 2x lysis buffer was added, followed by 1x lysis buffer to a total volume of 40 ml. The cells were resuspended by agitating and disrupted by sonication. Nucleic acids and cell debris were removed by adding 0.15% PEI (polyethyleneimine) from a 5% (w/v) stock, then centrifuged for 60 minutes at 16,000 rpm. The supernatant was further clarified by filtration (Acrodisc filters, 0.2 µm).
Column 1: 5-ml HisTrap Crude FF column (GE Healthcare)
Column 2: Gel filtration, Hiload 16/60 Superdex S200 prep grade, 120 ml (GE Healthcare) |
Solutions:
Lysis buffer: 50 mM HEPES, pH 7.5, 500 mM NaCl, 10 mM imidazole, 5% glycerol, 0.5 mM TCEP.
Wash buffer: 50 mM HEPES, pH 7.5, 500 mM NaCl, 30 mM imidazole, 5% glycerol, 0.5 mM TCEP.
Elution buffer: 50 mM HEPES, pH 7.5, 500 mM NaCl, 300 mM imidazole, 5% glycerol, 0.5 mM TCEP.
Gel Filtration Buffer - 10mM HEPES pH 7.5, 500mM NaCl, 5% glycerol and 0.5 mM TCEP
|
Procedure: The clarified supernatant was loaded on a 5-ml HisTrap Crude FF column at 4 ml/min using an AKTAxpress system. The column was washed with wash buffer containing 30 mM imidazole, and eluted with elution buffer containing 300 mM imidazole. The eluted fraction was collected automatically and further fractionated on a S200 HR 16/60 gel filtration column equilibrated with gel filtration buffer. Fractions containing ADAA protein were identified by SDS-PAGE, combined, and concentrated to 15 mg/ml using centrifugation (5 kDa MWCO, Centricon). Concentrated protein samples were flash frozen in liquid nitrogen and stored at -80 °C |
Enzymatic treatment: His-tagged TEV protease was added to the protein using a 1:20 TEV to protein ratio (mg/mg). It was incubated overnight at 4°C. |
Column 3:Ni-NTA (Qiagen) |
Buffers:
Wash buffer: 50 mM HEPES, pH 7.5, 500 mM NaCl, 30 mM imidazole, 5% glycerol, 0.5 mM TCEP.
Elution buffer: 50 mM HEPES, pH 7.5, 500 mM NaCl, 300 mM imidazole, 5% glycerol, 0.5 mM TCEP. |
Procedure: Following TEV digestion, the protein sample was passed through 1ml Ni-NTA in a 10mm gravity flow column and the flow-through collected. The column was washed with 10 ml of wash buffer and the wash and flow-through fractions were analyzed by SDS-PAGE and pooled. |
Mass spectrometry characterization : ESI-MS revealed that the protein had a mass of 41200Da (Expected mass 41330Da), the -130Da mass difference was attributed to loss of the initiator methionine. |
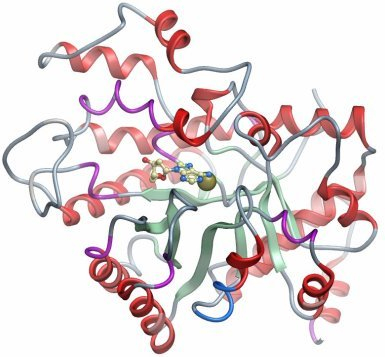
Compound: Adenine deoxyriboside (deoxyadenosine) |
Protein concentration: Protein stored at -80°C was thawed and diluted with buffer (10 mM HEPES pH7.5, 150 mM NaCl). A 2 to 3 times molar excess of compound was mixed with the protein and the solution was concentrated to 25 mg/ml using a Centricon filtration unit with a 5kDa MW cut-off. |
Crystallization: Crystals were grown at 20°C by vapour diffusion in sitting drops by mixing protein (25 mg/ml) and well solution containing 20% PEG 3350 and 0.20M NaNO3 at a protein to precipitant ratio of 1:2. A crystal was cryo-protected using well solution supplemented with 25% (v/v) glycerol and flash-cooled in liquid nitrogen.
|
| Data Collection: Resolution (scaled): 1.6 Å; X-ray source: rotating anode (Rigaku FR-E SuperBright) |