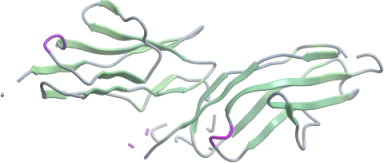
MALT1
PDB:3K0W
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:BC030143
Entry Clone Source:MGC
SGC Clone Accession:malt1.129.326.SDC125H02 (SDC125H02)
Tag:N-terminal tag: MGSSHHHHHHSSGLVPRGS
Host:
Construct
Prelude:
Sequence:mgsshhhhhhssglvprgsKITVNPESKAVLAGQFVKLCCRATGHPFVQYQWFKMNKEIPNGNTSELIFNAVHVKDAGFYVCRVNNNFTFEFSQWSQLDVCDIPESFQRSVDGVSESKLQICVEPTSQKLMPGSTLVLQCVAVGSPIPHYQWFKNELPLTHETKKLYMVPYVDLEHQGTYWCHVYNDRDSQDSKKVEIIIDELNNLGHPDNKEQTTDQ
Vector:
Growth
Medium:
Antibiotics:
Procedure:Competent BL-21(DE3) cells (Invitrogen, C6000-03) were transformed and grown using the LEX system (HarbingerBiotech) at 37 °C in 2L bottles (VWR, 89000-242) containing 1800 ml of TB (Sigma T0918) supplemented with 150 mM glycerol, 100 µM Kanamycin and 600 µl antifoam 204 (Sigma A-8311). At OD600 = 6, the temperature was reduced to 15 °C, and one hour later the culture was induced with 100 µM IPTG (BioShop IPT001) and incubated overnight (16 hours) at 15 °C. Cell pellets were collected by centrifugation (12,227 xg, 20 mins) and frozen in liquid nitrogen.
Purification
Procedure
Cleared lysate was rocked with TALON metal-affinity resin (BD Biosciences, 635504) (1.5 mL settled beads per L cell culture) at 4 °C. Resin was transferred to a column and washed with 5 column volumes (cv) of Wash buffer A, 5 cv of Wash buffer B, and 5 cv of Wash buffer A. The protein was eluted with 2 cv of Elution buffer. The protein was further purified by gel filtration through a HighLoad 16/60 Superdex 200 column (GE Healthcare, 17-1069-01) equilibrated with Gel Filtration buffer. Fractions containing protein (analyzed by ABS280 nm) were pooled and concentrated to 0.5-1.0 mM using concentrators (Amicon, UFC901024) with 5 kDa cutoff. The yield of the protein was approximately 20 mg per L of bacterial culture. Coomassie-stained, SDS-PAGE showed that the product was pure and Mass-spectroscopy by LCMS (Agilent 1100 Series) showed that the protein has 130 Da more than the calculated molecular weight
Extraction
Procedure
Cell pellets were resuspended in Lysis buffer(30 mL per L culture), lysed using a Microfluidizer (Microfluidics, M110-EH) at 18,000 psi, and cleared by centrifugation (40,000 xg for 30 minutes).
Concentration:
Ligand
MassSpec:
Crystallization:Protein and Bcl-10 peptide were mixed in molor ratio 1:2, and incubated overnight at 4 degC. Crystals were grown at 18 degC using the sitting drop method in Intelli-plates (Art Robbins, 102-0004-00) by mixing equal volumes of protein (10 mg/ml) and Crystallization Buffer (1.5 M MgCl2, 0.1 M Bicine, pH 9.0). Suitable crystals were cryoprotected by immersion in well solution supplemented with 15% (v/v) glycerol prior to dunking and storage in liquid nitrogen.
NMR Spectroscopy:
Data Collection:Diffraction data from a crystal of the tandem Ig domains of Malt1was collected at beamline CMCF-1 at the Canadian Light Source. The data set was integrated and scaled using the HKL2000 program package.
Data Processing:The structure was solved by molecular replacement techniques using the program PHASER and search model PDB entry 3BFO. The C-terminal Ig domain, identical to that of 3BFO was located using Phaser, while the N-terminal Ig domain (a modified version of 3BFO in which the sequence was changed to match that of the N-terminal domain using the JCSG SCWRL server) was manually positioned into the difference density of the initial electron density maps. Iterative manual model building using the graphics program Coot and maximum-likelihood, maximum-entropy, and TLS refinement with the program BUSTER-TNT led to a model with an R factor of 21.83% (Rfree 24.89%) for data between 40.47-2.80 Å. Parameters for Translation/liberation/screw (TLS) refinement were generated using the TLSMD web server.