CENTA1
PDB:3LJU
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:BC033747
Entry Clone Source:MGC AT53-B3
SGC Clone Accession:HPC077-H02
Tag:mhhhhhhssgrenlyfq*g
Host:BL21-CodonPlus(DE3)-RIL
Construct
Prelude:Tag not removed
CENTA1:K3-F370
Sequence:mhhhhhhssgrenlyfqgKERRRAVLELLQRPGNARCADCGAPDPDWASYTLGVFICLSCSGIHRNIPQVSKVKSVRLDAWEEAQVEFMASHGNDAARARFESKVPSFYYRPTPSDCQLLREQWIRAKYERQEFIYPEKQEPYSAGYREGFLWKRGRDNGQFLSRKFVLTEREGALKYFNRNDAKEPKAVMKIEHLNATFQPAKIGHPHGLQVTYLKDNSTRNIFIYHEDGKEIVDWFNALRAARFHYLQVAFPGASDADLVPKLSRNYLKEGYMEKTGPKQTEGFRKRWFTMDDRRLMYFKDPLDAFARGEVFIGSKESGYTVLHGFPPSTQGHHWPHGITIVTPDRKFLFACETESDQREWVAAFQKAVDRPMLPQEYAVEAHF
Vector:pET28-mhl (GI:134105571)
Growth
Medium:Terrific Broth
Antibiotics:Kanamycin 50 µg/mL Chloramphenicol 25 µg/mL
Procedure:LEX Bubbling. The target protein was expressed in E. coli by inoculating 100 mL of overnight culture grown in Luria-Bertani medium into 2L Terrific Broth medium in the presence of 50 µg/mL kanamycin and 50 µg/mL chloramphenicol at 37 °reeC. When OD600 reached ~3.0, the temperature of the medium was lowered to 18 µC and the culture was induced with 1 mM IPTG. The cells were allowed to grow overnight before they were harvested and flash frozen in liquid nitrogen and stored at -80 °C.
Purification
ProcedureThe lysate was centrigued at 15,000 rpm for 45 minutes and the supernatants were mixed with 1.5 mL 50% Ni-NTA beads, and incubated at 4 °reeC for 1.5 hours. The supernatant was then passed through a gravity column (Poly-Prep, Bio-Rad, Catalog #731-1550) and the beads were washed using 10 mL washing buffer twice. The protein bound to beads were eluted using 10 mL elution buffer twice. The flow-through was collected and loaded onto Supderdex-200 gel filtration column. Eluted fractions were pooled and concentrated using amicon centrifugal filter (m.w. cut-off 10,000 ). The purity of the proteins was higher than 95% judged by SDS-PAGE.
Extraction
ProcedureFrozen cells were thawed and resuspended in 80 mL extraction buffer with freshly added 1mM PMSF/Benzomidine, 5U/ml of Benzonase (Sigma Catalog # E1014, 250U/µL), 0.5% CHAPS, and supplemented with protease inhibitor cocktail (SIGMA Catalog # P8849), and lysed using microfludizer(17000 psi).
Concentration:25.6 mg/mL (stock)
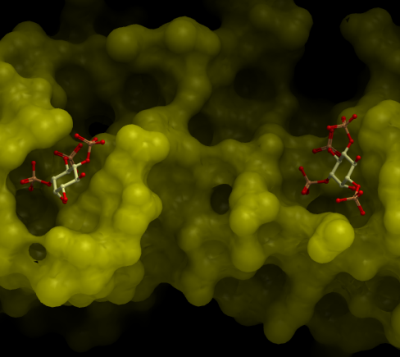
LigandZn, IP4
MassSpec:Native: 44928.82, expected 44925.78
Crystallization:Buffer for protein is 20mM HEPES pH7.3, 300mM NaCl, 1mM TCEP, 5% Glycerol.
Protein was mixed with 2.0mg/mL PIP3: (L-alpha-Phosphatidyl-D-myo-inositol 3,4,5-triphosphate, dioctanoyl, from SIGMA, catalog no. P9953-.1mg Lot# 126K1035, Formula: C25H50O22P4, F.W. 838.6, at 1:1 ratio molarity ratio) and then was setup in sitting drops with Red Wings and SGC-I screens at a size of 0.3uL protein:0.3uL well solution drops. Plate crystals were seen in conditions RW: G02, H03, D08, G09 in two days, more crystals are seen in conditions RW: B11, E08, G11, C03 in 2-3 weeks time.
Crystal used for data collection was grown in RW:C3 (20.0% P3350, 0.2M NH4Cl, no buffer), cryo used paratone.
It is not clear whether the side chain of PIP3 is not visible in the electron density map or residual IP4 in the PIP3 reagent binds to CENTA1 tighter such that IP4 crystalizes with CENTA1 instead of PIP3.
NMR Spectroscopy:
Data Collection:
Data Processing: