BMPR1B (ALK6) belongs to the bone morphogenetic protein (BMP) receptor family of transmembrane serine/threonine kinases. Secreted BMPs, which are members of the TGF-beta superfamily involved in endochondral bone formation and embryogenesis, induce heteromeric assembly of type II and type I receptors. Five members of the type II and seven members of the type I receptor (also known as activin-like kinases, ALK1-7) are identified in mammals (Sebald et al., 2004). The type II receptor functions to activate the type I receptor by phosphorylation of its glycine-serine rich (GS) domain. Subsequently, the type I receptor binds and phosphorylates members of the SMAD family of transcription factors. BMPR1B binds preferentially to GDF-5 but also accepts BMP2 and BMP7 as ligand (Nickel et al., 2005), (Sebald et al., 2004).
Knockout studies reveal significant functional redundancy between Bmpr1a and Bmpr1b, but some physiological processes specifically require Bmpr1b. Mice lacking Bmpr1b exhibit defects in the appendicular skeleton. These mice show reduced proliferation of prechondrogenic cells and defective chondrocyte differentiation in the phalangeal region. Consequently, the adult mice lack the proximal interphalangeal joint and phalanges are replaced by a single rudimentary element (Yi et al., 2000), (Yoon et al., 2005). Additional neurological features include elevated apoptosis at the end of neurogenesis and the failure of a specific subpopulation of axons to enter the optic nerve head (Liu et al., 2003).
Mutations in the human BMPR1B gene cause type A2 or type C brachydactyly, characterised by shortening and lateral deviation of fingers and toes to a variable degree (Lehmann et al., 2003). A deletion in BMPR1B has also been identified in acromesomelic chondrodysplasia with genital anomalies, a rare hereditary skeletal disorder characterized by short stature, very short limbs, and hand/foot malformations underlining the central role of BMPR1B in chondrogenesis and bone morphogenesis (Demirhan et al., 2005).
Dysregulation of BMPR1B has been found in several cancer types. Reduced expression correlates with poor prognosis in prostate and breast cancers (Miyazaki et al., 2004), (Bokobza et al., 2009). A SNP in a miR-125b target site of the 3' untranslated region of BMPR1B is also associated with breast carcinogenesis (Saetrom et al., 2009). Conversely, upregulated expression has been found in malignant glioma tumours (Yamada et al., 1996).
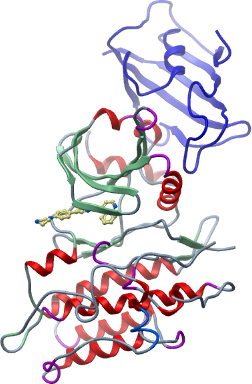
Here we present the structure of the GS and kinase domains of BMPR1B in complex with FKBP12 and the kinase inhibitor LDN-193189 (Yu et al., 2008), (Cuny et al., 2008) refined at 2.05 Å resolution.