Entry Clone Source: Site-directed mutagenesis |
Entry Clone Accession: n/a |
SGC Construct ID: PCTK1A-c031 |
GenBank GI number: gi|5453860 |
Vector: pNIC-CH. Details [ PDF ]; Sequence [ FASTA ] or [ GenBank ]
|
Amplified construct sequence:
TTAAGAAGGAGATATACTATGGAGACCTAC
ATTAAGCTGGACAAACTGGGCGAGGGTACC
TATGCCACCGTCTACAAAGGCAAAAGCAAG
CTCACAGACAACCTTGTGGCACTCAAGGAG
ATCAGACTGGAACATGAAGAGGGGGCACCC
TGCACCGCCATCCGGGAAGTGTCCCTGCTC
AAGGACCTCAAACACGCCAACATCGTTACG
CTACATGACATTATCCACACGGAGAAGTCC
CTCACCCTTGTCTTTGAGTACCTGGACAAG
GACCTGAAGCAGTACCTGGATGACTGTGGG
AACATCATCAACATGCACAACGTGAAACTG
TTCCTGTTCCAGCTGCTCCGTGGCCTGGCC
TACTGCCACCGGCAGAAGGTGCTACACCGA
GACCTCAAGCCCCAGAACCTGCTCATCAAC
GAGAGGGGAGAGCTCAAGCTGGCTGACTTT
GGCCTGGCCCGAGCCAAGTCAATCCCAACA
AAGACATACGACAATGAGGTGGTGACACTG
TGGTACCGGCCCCCTGACATCCTGCTTGGG
TCCACGGACTACTCCACTCAGATTGACATG
TGGGGTGTGGGCTGCATCTTCTATGAGATG
GCCACAGGCCGTCCCCTCTTTCCGGGCTCC
ACGGTGGAGGAACAGCTACACTTCATCTTC
CGTATCTTAGGAACCCCAACTGAGGAGACG
TGGCCAGGCATCCTGTCCAACGAGGAGTTC
AAGACATACAACTACCCCAAGTACCGAGCC
GAGGCCCTTTTGAGCCACGCACCCCGACTT
GATAGCGACGGGGCCGACCTCCTCACCAAG
CTGTTGCAGTTTGAGGGTCGAAATCGGATC
TCCGCAGAGGATGCCATGAAACATCCATTC
TTCCTCAGTCTGGGGGAGCGGATCCACAAA
CTTCCTGACACTACTTCCATATTTGCACTA
AAGGAGATTCAGCTACAAAAGGAGGCCAGC
CTTCGGTCTGCGCACCATCATCACCACCAT
T
|
Expressed protein sequence:
METYIKLDKLGEGTYATVYKGKSKLTDNLV
ALKEIRLEHEEGAPCTAIREVSLLKDLKHA
NIVTLHDIIHTEKSLTLVFEYLDKDLKQYL
DDCGNIINMHNVKLFLFQLLRGLAYCHRQK
VLHRDLKPQNLLINERGELKLADFGLARAK
SIPTKTYDNEVVTLWYRPPDILLGSTDYST
QIDMWGVGCIFYEMATGRPLFPGSTVEEQL
HFIFRILGTPTEETWPGILSNEEFKTYNYP
KYRAEALLSHAPRLDSDGADLLTKLLQFEG
RNRISAEDAMKHPFFLSLGERIHKLPDTTS
IFALKEIQLQKEASLRSAHHHHHH
Engineered S319D phospho-mimetic mutation shown bold and underlined. |
Host: BL21(DE3)-R3-pRARE2
|
Growth medium, induction protocol: A glycerol stock was used to inoculate a 10ml starter culture containing LB media with 50µg/ml Kanamycin and 34 µg/ml chloramphenicol. The starter culture was grown overnight at 37°C with shaking at 200 rpm. The following morning, four flasks containing 1 L LB/kanamycin/chloramphenicol were each inoculated with 5 ml of the starter culture. Cultures were incubated at 37°C with shaking at 180 rpm until an OD600nm ≥ 0.7 was reached. The flasks were then cooled down to 18°C and 0.5mM IPTG added to induce protein expression overnight. Cells were harvested by centrifugation at 4500 rpm at 4°C for 15 min. Cell pellets from each flask were resuspended in 30ml binding buffer (50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole), transferred to 50 ml tubes, and stored at -20°C. |
Extraction buffer, extraction method: The frozen cells were thawed and 0.5mM TCEP, 1mM PMSF added to the cell suspension. The cells were lysed by ultrasonication over 12 min with the sonicator pulsing ON for 5 sec and OFF for 15 sec. The cell lysate was spun down by centrifugation at 17000 rpm at 4°C for 1 h. The supernatant was recovered for purification. |
Column 1: Ni-Affinity Chromatography: 5ml of 50 % Ni-sepharose slurry (Amersham) was applied onto a 1.5 x 10 cm column. The column was first washed with deionised distilled H2O, and then equilibrated with binding buffer. |
Column 1 Buffers:
Binding buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole
Wash buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 30mM Imidazole
Elution buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 50 to 250mM Imidazole |
Column 1 Procedure: The supernatant was applied by gravity flow onto the Ni-sepharose column. The bound protein was eluted by applying a step gradient of imidazole (5 ml fractions of elution buffer supplemented with 50mM, 100mM, 150mM and 250mM imidazole). 10mM DTT was added to each fraction collected for overnight storage at 4°C. |
Enzymatic treatment:: N/A |
Column 2: Size Exclusion Chromatography - S200 HiLoad 16/60 Superdex run on ÄKTA-Express. |
Column 2 Buffers: Gel Filtration buffer: 300mM NaCl, 50mM HEPES pH 7.5, 0.5mM TCEP |
Column 2 Procedure: Prior to applying the protein, the S200 16/60 column was washed and equilibrated with gel filtration buffer. Eluted protein from the Ni-sepharose column was pooled and concentrated to 3ml using an Amicon Ultra-15 filter with a 10kDa cut-off. The concentrated protein was directly applied onto the equilibrated S200 16/60 column, and run at a flow-rate of 1 ml/min. Fractions containing the protein were pooled together, and 10mM DTT was added for overnight storage at 4°C. |
Column 3: Anion Exchange Chromatography (monoQ) |
Column 3 Buffers:
Buffer A: 50mM TRIS, pH 9.0
Buffer B: 50mM TRIS, pH 9.0; 1M NaCl |
Column 3 Procedure: PCTK1 protein from gel filtration was buffer exchanged into 50mM Tris pH 9.0 and loaded onto a 1 ml monoQ anion exchange column equilibrated in the same buffer. A linear elution gradient was run from 0-1M NaCl. PCTK1 containing fractions were pooled and the buffer adjusted to 25 mM HEPES, pH 7.5, 250 mM NaCl, 5 % Glycerol, 10 mM DTT during concentration in a 5 kD MWCO Amicon Ultra concentrator. |
Concentration: The protein was concentrated in a 5 kD MWCO Amicon Ultra concentrator to 15 mg/ml using an estimated extinction coefficient of 35870 and MW of 37252. |
Mass spectrometry characterization: The purified native protein was homogeneous and had an experimental mass of 37252.74 (expected MW = 37252). Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. |
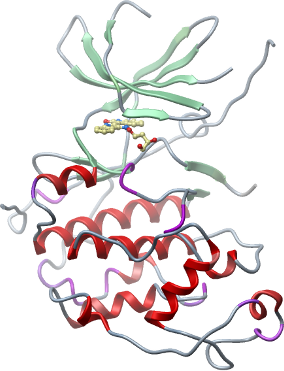
Crystallisation: Protein was buffered in 25 mM HEPES, pH 7.5, 250 mM NaCl, 5 % Glycerol, 10 mM DTT. Protein was concentrated to 15 mg/ml in the presence of indirubin E804 (final inhibitor concentration of 1 mM). Crystals were grown by micro-seeding at 20°C in 130 nl sitting drops mixing 96 nl protein solution with 10 nl micro-seed solution (crystals prepared from the same precipitant) and 24 nl of a reservoir solution containing 2.1M Na-formate pH 7.0, 0.1 M Bis-Tris pH 7.0. On mounting crystals were cryo-protected with reservoir solution mixed with 25% glycerol. |
Data Collection: Resolution: Resolution: 2.4 Å, X-ray source: Diamond I03. The native crystal diffracted to a resolution of 2.4 Å. |