Entry Clone Source: MGC |
Entry Clone Accession: IMAGE:2822884 |
SGC Construct ID: DHRS4A-c601 |
GenBank GI number: gi|4105190 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
CATATGCACCATCATCATCATCATTCTTCT
GGTGTAGATCTGGGTACCGAGAACCTGTAC
TTCCAATCCATGGCCAGCTCCGGGATGACC
CGCCGGGACCCGCTCGCAAATAAGGTGGCC
CTGGTAACGGCCTCCACCGACGGGATCGGC
TTCGCCATCGCCCGGCGTTTGGCCCAGGAC
GGGGCCCATGTGGTCGTCAGCAGCCGGAAG
CAGCAGAATGTGGACCAGGCGGTGGCCACG
CTGCAGGGGGAGGGGCTGAGCGTGACGGGC
ACCGTGTGCCATGTGGGGAAGGCGGAGGAC
CGGGAGCGGCTGGTGGCCACGGCTGTGAAG
CTTCATGGAGGTATCGATATCCTAGTCTCC
AATGCTGCTGTCAACCCTTTCTTTGGAAGC
ATAATGGATGTCACTGAGGAGGTGTGGGAC
AAGACTCTGGACATTAATGTGAAGGCCCCA
GCCCTGATGACAAAGGCAGTGGTGCCAGAA
ATGGAGAAACGAGGAGGCGGCTCAGTGGTG
ATCGTGTCTTCCATAGCAGCCTTCAGTCCA
TCTCCTGGCTTCAGTCCTTACAATGTCAGT
AAAACAGCCTTGCTGGGCCTGACCAAGACC
CTGGCCATAGAGCTGGCCCCAAGGAACATT
AGGGTGAACTGCCTAGCACCTGGACTTATC
AAGACTAGCTTCAGCAGGATGCTCTGGATG
GACAAGGAAAAAGAGGAAAGCATGAAAGAA
ACCCTGCGGATAAGAAGGTTAGGCGAGCCA
GAGGATTGTGCTGGCATCGTGTCTTTCCTG
TGCTCTGAAGATGCCAGCTACATCACTGGG
GAAACAGTGGTGGTGGGTGGAGGAACCCCG
TCCCGCCTCTGACAGTAAAGGTGGATACGG
ATCCGAA
|
Final protein sequence (tag sequence in lowercase)
mhhhhhhssgvdlgtenlyfqsMASSGMTR
RDPLANKVALVTASTDGIGFAIARRLAQDG
AHVVVSSRKQQNVDQAVATLQGEGLSVTGT
VCHVGKAEDRERLVATAVKLHGGIDILVSN
AAVNPFFGSIMDVTEEVWDKTLDINVKAPA
LMTKAVVPEMEKRGGGSVVIVSSIAAFSPS
PGFSPYNVSKTALLGLTKTLAIELAPRNIR
VNCLAPGLIKTSFSRMLWMDKEKEESMKET
LRIRRLGEPEDCAGIVSFLCSEDASYITGE
TVVVGGGTPSRL |
Host: BL21(DE3)-R3-pRARE2 |
Tags and additions: N-terminal hexa-Histidine-tag with TEV protease cleavage site |
Expression: BL21(DE3)-R3 glycerol stock harbouring the DHRS4-pNIC28-Bsa4 plasmid was inoculated into 50ml of TB with 100µg/ml of kanamycin and 34µg/ml chloramphenicol and grown overnight at 37°C, 200 rpm. 10ml of overnight culture were added to 1L X 6 of TB with 100µg/ml ampicilin and incubated at 37°C, 160rpm. After the OD 600 reached 1.0, the temperature was dropped to 18°C and 1ml of 0.2M IPTG was added to the final concentration of ~0.2mM. The culture was then incubated with shaking overnight at 18°C, 160rpm. The following morning the 6L culture was harvested and centrifuged for 10min at 6000rpm. Supernatant was discarded and cell pellets were resuspended in 80ml of lysis buffer and frozen at -80°C. |
Extraction: The thawed cells were broken by a high pressure homogenizer. 5 ul Benzonase and 0.15% polyethyleneimine (PEI) was added to the lysate. The lysate was centrifuged at 16,000 rpm for 45 minutes and the supernatant collected for purification.
|
Purification:
Column 1: Ni-affinity chromatography. Ni-NTA, 5 ml of 50% slurry in column, washed with binding buffer.
Column 2: Size Exclusion Chromatography. Superdex S200 16/60 HiLoad , equilibrated with in GF buffer
|
Buffers:
Lysis Buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 5% Glycerol, 10mM Imidazole 0.5 mM TCEP, protease inhibitors
Wash buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 20 mM Imidazole , 5% Glycerol, 0.5 mM TCEP.
Elution buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 250 mM Imidazole , 5% Glycerol, 0.5 mM TCEP.
Gel filtration (GF) buffer: 10 mM HEPES pH 7.5, 500 mM NaCl, 5% Glycerol, 0.5 mM TCEP. |
Procedure: The clarified lysate was loaded by gravity flow on the Ni-NTA column. The column was then washed with 20 ml lysis buffer, followed by 20ml wash buffer. The protein was eluted by gravity flow by applying 20ml of elution buffer and collected in 2ml fractions. The fractions were analyzed by SDS-PAGE, pooled together, concentrated and applied to the gel filtration column, pre-equilibrated with GF buffer. The absorbance at 280nm was monitored and fractions were collected and analyzed by SDS-PAGE. Protein fractions were pooled for TEV cleavage.
|
TEV cleavage: The His-tag was cleaved with 1mg TEV per 40mg target protein at 4°C overnight. The protein was further purified on Ni-NTA equilibrated with GF buffer using buffers as above.
|
Concentration and buffer exchange: DHRS4 was diluted with 10mM HEPES, 5% glycerol to reduce [NaCl] to 250mM and then concentrated to 18.5 mg/ml using Amicon Ultra-15 concentrators with 10kDa cutoff.
|
Mass spectrometry characterization: The calculated mass of the tagged and TEV cleaved protein was calculated to be 30124.6 and 27658.9, respectively. This was experimentally confirmed by mass spectrometry analysis.
|
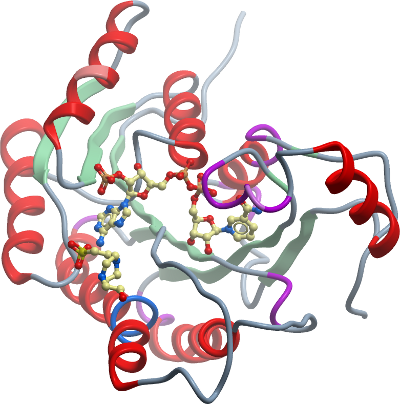
Crystallisation with NADPH
Prior to crystallization the protein was supplemented with 6.7 mM NADPH. Crystals were grown at 20°C in 150nl sitting drops mixing DHRS4 (18.5mg/ml) with and reservoir solution containing 0.1M potassium thiocynate and 30% w/v PEG MME 2000 in a 1:2 ratio. Crystals were cryo-protected in reservoir solution containing 25% (v/v) ethylene glycol, and flash-cooled in liquid nitrogen. |
Data collection: Resolution: 1.9 Å; X-ray source: Diamond Light Source, beamline IO3 |