Calcium dependent protein klinases, absent in animals but found in apicomplexan parasites and specific plants, typically consist of a kinase domain with a calcium-binding CDPK Activation Domain (CAD) in the C-terminus. Often, the CAD is made up of a long flexible helix (CH1) followed by two pairs of EF-hands linked by another flexible helix (CH2). In Plasmodium vivax, the parasite responsible for infecting humans with a lethal strain of malaria, there are four known CDPKs in the genome, including PvCDPK3, or PVX_119610 (PlasmoDB ID).
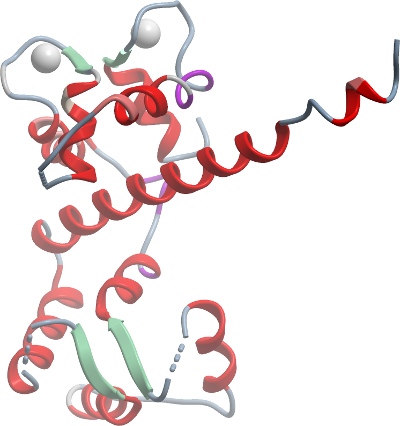
Using the activated and inactivated structures of CDPKs from Toxoplasma and Cryptosporidium (3HZT, 3HX4, 3IGO), we have previously proposed that a model for the activation of CDPKs based on calcium-triggered refolding of the CAD from the state exemplified in 3HXT to the conformation in 3IGO [1, 2], concurrent with the translocation of this domain from the substrate-blocking position to the "opposite" side of the kinase domain. Here, we present the structure of the CAD domain alone from PfCDPK3, which provides a deeper understanding of the mechanism of refolding.
The CAD domain forms a dimer in solution in the absence of the kinase domain. For the most part, this domain is folded in the same manner as it is in the activated full length CDPK structure (3HX4, 3IGO) and the CAD domain of the Pf orthology (3K21). The major difference here is that the CH1 helix, in the absence o the kinase domain, is unbent, unlike in the full-length structures where it is bent and wrapped around the EF hands. This enables us to speculate about the process of how the CAD refolds [2].