Hsp90 is a ubiquitous chaperone found in all characterized eukaryotes. In humans, Hsp90
is heavily researched as a target for diseases such as cancer and diabetes. Consequently,
there are a number of known inhibitors, ranging from natural products to synthetic
compounds. Some of these compounds have been reported to have potent anti-parasitic
effects.
In Trypansoma parasites, the orthologues to Hsp90 is TbHsp83 (the naming scheme is
based on the variable molecular weight of the chaperone in different organisms). We
conducted an experiment to screen a panel of known or suspected Hsp90 inhibitors
against TbHsp83 and found a number of them to bind with high affinity. One such
compound is BIIB021, a drug candidate developed by Conforma Therapeutics that went
into phase II clinical trial against gastrointestinal stromal tumor. It has also gone into
phase I trial against lymphoma.
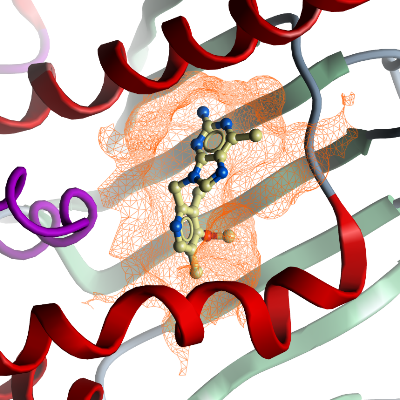
We have crystallized the N-terminal ATPase domain of TbHsp83 with BIIB021 bound.
Comparing this structure to our other TbHsp83 structures with other compounds bound
(PDB ID: 3OMU, 3OPD) shows that domain, similar to that
in the human orthologues (PDB ID: 3QDD), is flexible and adopts structurally to its
inhibitors.