CBP is a central protein in a variety of biological processes including cell growth and transformation, genomic stability, development, neuronal plasticity and memory formation as well as energy homeostasis. The fundamental role of CBP is reflected in the phenotype of the Cbp knockout: homozygous mutant mice die in utero with signs of defective blood vessel formation in the central nervous system, and developmental retardation as well as delays in both primitive and definitive hematopoiesis (Kalkhoven, 2004).
CBP acts as a general transcriptional coactivator and acetylates histones as well as non-histone proteins. Acetylation of non-histone proteins can have a positive or negative effect on transcriptional regulation by affecting protein-protein interaction, protein-DNA interaction, nuclear retention or protein half-life of these molecules (Kalkhoven, 2004).
Mutations in the CBP gene can lead to Rubinstein-Taybi syndrome (RTS), a rare human genetic disorder characterized by mental retardation and physical abnormalities; many patients with RTS either have breakpoints or microdeletions in chromosome 16p13.3 where the CBP gene is located but also heterozygous point mutations can lead to RTS (Rouaux et al., 2004). CBP plays also a role in the etiology of other human neurological disordes such as Amyotrophic lateral sclerosis, Alzheimer's disease and also in poly glutamine diseases, inflammatory diseases and several cancer forms (Matt, 2002; Hallam and Bourtchouladze, 2006).
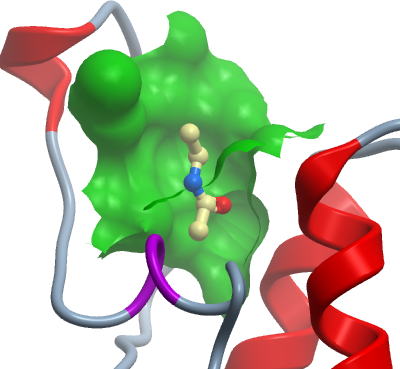
CBP is a multidomain protein consisting of three cysteine-histidine-rich domains (CH1, CH2, CH3) serving as docking modules for numerous transcriptional regulators, the binding site for the CREB transcription factor (KIX domain), a bromodomain, two PHD type zinc finger motifs (LAP fingers, TTC fingers), an N-terminal nuclear receptor binding domain, a C-terminal glutamine-rich domain and a histone acetyltransferase (HAT) domain (Blobel, 2002). In order to enable the development of potent CBP inhibitor that target the bromodomain we solved co-crystal structures of the CBP bromodomains with acetyl lysine as well as complexes with three fragments that act as acetyl lysine competitive inhibitors. These molecules include N-Methyl-2-pyrrolidone (NMP), dimethylsulfoxide (DMSO) and a substituted hydroquinazolin.