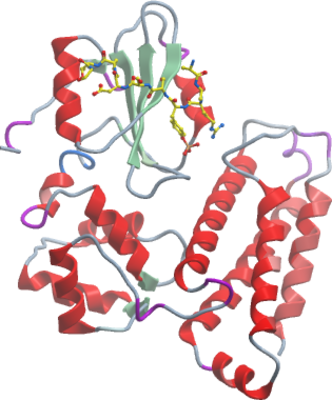
The Cbl (for Casitas B-lineage lymphoma) family of E3 ubiquitin ligases comprises the three members Cbl, Cbl-b and Cbl-c. The founding member Cbl was discovered first as the oncogenic protein v-Cbl, a Gag-fusion transforming protein of Cas NS-1 retrovirus, which causes pre- and pro-B lymphomas in mice. Both Cbl (or c-Cbl) and Cbl-b are widely expressed and share a common protein as well as gene structure, whereas Cbl-c shows only sequence identity to the amino-terminal sequences of Cbl and Cbl-b, but lacks the leucine zipper motif and most of the proline-rich domain in the C-terminus. The N-terminus of the Cbl proteins is composed of a tyrosine kinase-binding (TKB) domain, also called phosphotyrosine binding (PTB) domain, a short linker region and the RING-type zinc finger. The TKB domain mediates interaction with other proteins and consists of a four-helix bundle (4H), a calcium-binding EF hand and a divergent SH2 domain (Swaminathan and Tsygankov, 2006), (Loeser and Penninger, 2007b).
Cbl-b plays a role in early hematopoietic development and is a negative regulator of T-cell receptor, B-cell receptor and high affinity immunoglobulin epsilon receptor signal transduction pathways (Loeser and Penninger, 2007a), (Qu et al., 2004), (Huang and Gu, 2008). Accordingly, knockout mice lacking Cbl-b show enhanced T-cell activation with hyperresponsiveness to T-cell receptor stimulation. Loss of Cbl-b in mice also uncouples T-cell activation from CD28 co-stimulation and makes these mice more susceptible to spontaneous and peptide induced activation pointing to a role of Cbl-b in promoting anergy, which prevents T-cells to react against self-antigens autoimmunity (Loeser and Penninger, 2007a), (Rudd and Schneider, 2000), (Mueller, 2004). Cbl-b thereby maintains the balance between tolerance, activation, and autoimmunity.
Outside the immune system Cbl-b negatively regulates insulin-like growth factor 1 signaling during muscle atrophy caused by unloading and is also involved in EGFR ubiquitination and internalization (Nakao et al., 2009), (Pennock and Wang, 2008).
Defects of Cbl-b are associated with a variety of human diseases. A genome wide association study has found variants of Cbl-b to be associated with multiple sclerosis and impairment of the Cbl-b signaling pathway is thought to contribute to human autoimmune diseases, including type 1 diabetes (Sanna et al., 2010), (Bergholdt et al., 2005). Loss of Cbl-b may be responsible for the craniofacial phenotype in patients with deletions of the proximal 3q region, a phenotype that is also found in the Cbl-b knockout mouse due to enhanced osteoclastogenesis in the bones of the skull (Simovich et al., 2008).
Here we report the crystal structure of the TKB domain of Cbl-b in complex with EGFR pY1069 peptide refined at 2.3 Å resolution.