Entry Clone Source: Synthetic |
Entry Clone Accession: n/a |
SGC Construct ID: BAZ2BA-c011 |
GenBank GI number: gi|7304923 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
TACTTCCAATCCATGAGCGTGAAAAA
ACCGAAACGCGATGATAGCAAAGATC
TGGCGCTGTGCAGCATGATTCTGACC
GAAATGGAAACCCATGAAGATGCGTG
GCCGTTTCTGCTGCCGGTGAACCTGA
AACTGGTTCCGGGCTATAAAAAAGTG
ATTAAAAAACCGATGGATTTTAGCAC
CATTCGTGAAAAACTGAGCAGCGGTC
AGTATCCGAACCTGGAAACCTTTGCG
CTGGATGTGCGTCTGGTGTTTGATAA
CTGCGAAACCTTTAACGAAGATGATA
GCGATATTGGCCGTGCCGGTCATAAC
ATGCGCAAATATTTTGAAAAAAAATG
GACCGATACCTTTAAAGTTAGCTGAC
AGTAAAGGTGGATA
|
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfq^smSV
KKPKRDDSKDLALCSMILTEMETHED
AWPFLLPVNLKLVPGYKKVIKKPMDF
STIREKLSSGQYPNLETFALDVRLVF
DNCETFNEDDSDIGRAGHNMRKYFEK
KWTDTFKVS
^ TEV cleavage site |
Tags and additions: Cleavable N-terminal His6 tag. |
Host: BL21 (DE3)R3-pRARE2 (Phage resistant strain).
|
Growth medium, induction protocol: 5ml from a 50 ml overnight culture containing 50µg/ml kanamycin and 34µg/ml chloramphenicol were used to inoculate each of two 1L cultures of LB containing 50µg/ml kanamycin and 34µg/ml chloramphenicol. Cultures were grown at 37°C until the OD600 reached ~0.5 then the temperature was adjusted to 18°C. Expression was induced overnight using 0.5 mM IPTG at an OD600 of 0.9. The cells were collected by centrifugation and the pellet re-suspended in binding buffer and frozen.
Lysis buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5 mM Imidazole; 5% Glycerol.
Extraction buffer, extraction method: Frozen pellets were thawed and fresh 0.5 mM TCEP, 1 mM PMSF added to the lysate. Cells were lysed by sonication. The lysate was centrifuged at 21,000rpm for 60 minutes and the supernatant collected for purification. |
Column 1: Ion exchange - Nucleic acid removal. DEAE cellulose (DE52, Whatman), 10g of resin in 2.5x20cm column. The resin was hydrated in 2.5M NaCl, then washed with 20ml binding buffer prior to loading the sample. |
Column 1 Buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 0.5 mM Imidazole. |
Column 1 Procedure: Supernatant was applied by gravity flow, followed by a wash with 50ml binding buffer. The column flow-through was collected. |
Column 2: Ni-affinity. Ni-sepharose (Amersham), 5 ml of 50% slurry in 1.5x10 cm column, washed with binding buffer. |
Column 2 Buffers:
Binding buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 5 mM Imidazole.
Wash buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 30 mM Imidazole.
Elution buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol, 50 to 250 mM Imidazole (step elution).
|
Column 2 Procedure: The flowthrough from column 1 was loaded by gravity flow on the Ni-sepharose column. The column was then washed with 30 ml wash buffer at gravity flow. The protein was eluted by gravity flow by applying 5ml portions of elution buffer with increasing concentration of imidazole (50 mM, 100 mM, 150 mM, 200 and 250 mM); fractions were collected until essentially all protein was eluted. 10 mM DTT was added for overnight storage. |
Enzymatic treatment: The N-terminal His tag was cleaved by treatment with TEV protease, overnight. |
Column 3: Size Exclusion Chromatography. Superdex S200 16/60 HiLoad. |
Column 3 Buffer: 50 mM HEPES, pH 7.5; 150 mM NaCl; 0.5 mM TCEP. |
Column 3 Procedure: BAZ2B was concentrated and applied to an S200 16/60 HiLoad gel filtration column equilibrated in 25 mM HEPES, pH 7.5; 150 mM NaCl, 0.5 mM TCEP using an ÄKTA express system. |
Mass spectrometry characterization: LC- ESI -MS TOF gave a measured mass of 13598.4 for this construct as predicted from the sequence of this protein. |
Protein concentration: Protein was concentrated to 10mg/ml using an Amicon 3kDa cut-off concentrator. |
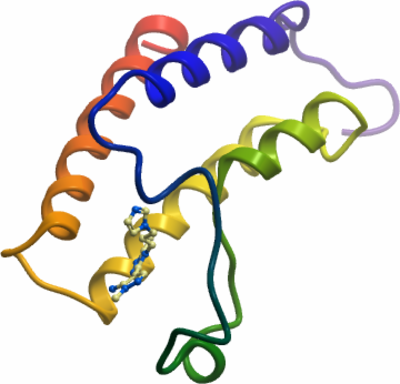
Crystallisation: Crystals grown at 4°C in E06639a inhibitor soaked. |
Data collection: Crystals were cryo-protected using the well solution supplemented by 25% ethylene glycol and flash frozen in liquid nitrogen.
X-ray source: Diffraction data were collected at 2.06Å from a single crystal on a Rigaku FR-E SuperBright.
Phasing: The structure was solved by molecular replacement using the BAZ2B apo-structure reported earlier. |