Entry Clone Source: TKC |
Entry Clone Accession: n/a |
SGC Construct ID: PIM1A-c001 |
GenBank GI number: gi|4505811 |
Vector: pLIC-SGC1. Details [PDF]; Sequence [FASTA] or [GenBank] |
Amplified construct sequence:
ATGCACCATCATCATCATCATTCTTC
TGGTGTAGATCTGGGTACCGAGAACC
TGTACTTCCAATCCATATGCTCTTGT
CCAAAATCAACTCGCTTGCCCACCTG
CGCGCCGCGCCCTGCAACGACCTGCA
CGCCACCAAGCTGGCGCCCGGCAAGG
AGAAGGAGCCCCTGGAGTCGCAGTAC
CAGGTGGGCCCGCTACTGGGCAGCGG
CGGCTTCGGCTCGGTCTACTCAGGCA
TCCGCGTCTCCGACAACTTGCCGGTG
GCCATCAAACACGTGGAGAAGGACCG
GATTTCCGACTGGGGAGAGCTGCCTA
ATGGCACTCGAGTGCCCATGGAAGTG
GTCCTGCTGAAGAAGGTGAGCTCGGG
TTTCTCCGGCGTCATTAGGCTCCTGG
ACTGGTTCGAGAGGCCCGACAGTTTC
GTCCTGATCCTGGAGAGGCCCGAGCC
GGTGCAAGATCTCTTCGACTTCATCA
CGGAAAGGGGAGCCCTGCAAGAGGAG
CTCGCCCGCAGCTTCTTCTGGCAGGT
GCTGGAGGCCGTGCGGCACTGCCACA
ACTGCGGGGTGCTCCACCGCGACATC
AAGGACGAAAACATCCTTATCGACCT
CAATCGCGGCGAGCTCAAGCTCATCG
ACTTCGGGTCGGGGGCGCTGCTCAAA
GACACCGTCTACACGGACTTCGATGG
GACCCGAGTGTATAGCCCTCCAGAGT
GGATCCGCTACCATCGCTACCATGGC
AGGTCGGCGGCAGTCTGGTCCCTGGG
GATCCTGCTGTATGATATGGTGTGTG
GAGATATTCCTTTCGAGCATGACGAA
GAGATCATCAGGGGCCAGGTTTTCTT
CAGGCAGAGGGTCTCTTCAGAATGTC
AGCATCTCATTAGATGGTGCTTGGCC
CTGAGACCATCAGATAGGCCAACCTT
CGAAGAAATCCAGAACCATCCATGGA
TGCAAGATGTTCTCCTGCCCCAGGAA
ACTGCTGAGATCCACCTCCACAGCCT
GTCGCCGGGGCCCAGCTAAAGTAAAG
GTGGATAC
|
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfq^smLL
SKINSLAHLRAAPCNDLHATKLAPGK
EKEPLESQYQVGPLLGSGGFGSVYSG
IRVSDNLPVAIKHVEKDRISDWGELP
NGTRVPMEVVLLKKVSSGFSGVIRLL
DWFERPDSFVLILERPEPVQDLFDFI
TERGALQEELARSFFWQVLEAVRHCH
NCGVLHRDIKDENILIDLNRGELKLI
DFGSGALLKDTVYTDFDGTRVYSPPE
WIRYHRYHGRSAAVWSLGILLYDMVC
GDIPFEHDEEIIGGQVFFRQRVSSEC
QHLIRWCLALRPSDRPTFEEIQNHPW
MQDVLLPQETAEIHLHSLSPGPS
^ TEV cleavage site |
Tags and additions: Cleavable N-terminal His6 tag. |
Host: BL21 (DE3)R3-pRARE2 (Phage resistant strain).
|
Growth medium, induction protocol: 5ml overnight culture in LB, 100µg/ml ampicillin were grown at 37°C and 0.5ml used to inoculate 1L of LB medium containing 100µg/ml ampicillin. Culture were grown at 37°C until the OD600 reached ~1.2 then cooled to 18°C. Expression was induced overnight using 0.5 mM IPTG. The cells were collected by centrifugation, transferred to 50ml tubes, resuspended in 30ml binding buffer and frozen.
Lysis buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% Glycerol.
Extraction buffer, extraction method: Frozen pellets were thawed on ice and 0.5 mM TCEP and 1 mM PMSF added. Cells were lysed using a high pressure cell disruptor. The lysate was centrifuged at 17,000rpm for 30 minutes. Supernatant was collected and binding buffer was added to 50ml. |
Column 1: Ion exchange - Nucleic acid removal. DEAE cellulose (DE52, Whatman), 10g of resin in 2.5x20cm column. The resin was hydrated in 2.5M NaCl, then washed with 20ml binding buffer prior to loading the sample. |
Column 1 Buffer:50 mM HEPES, pH 7.5; 500 mM NaCl; 5% Glycerol. |
Column 1 Procedure: Supernatant was applied by gravity flow, followed by a wash with 40ml binding buffer. The column flow-through was collected. |
Column 2: Ni-affinity. Ni-sepharose (Amersham), 5 ml of 50% slurry in 1.5x10 cm column, equilibrated with binding buffer. |
Column 2 Buffers:
Binding buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol.
Wash buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 20 mM Imidazole.
Elution buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol, 50 to 250 mM Imidazole (step elution).
|
Column 2 Procedure: The flowthrough from column 1 was loaded by gravity flow on the Ni-sepharose column. The column was then washed with 50 ml wash buffer at gravity flow. The protein was eluted by gravity flow by applying 5ml portions of elution buffer with increasing concentration of imidazole (50 mM, 100 mM and 250 mM); fractions were collected until essentially all protein was eluted. After elution DTT was added to a final concentration of 10 mM. |
Enzymatic treatment:Dephosphorylation (a GST fusion with the lambda phosphatase) and TEV protease cleavage. Samples containing Pim1 were pooled and treated with lambda phosphatase and TEV protease overnight at 4°C Protein was kept in elution buffer with the addition of 10 mM DTT and 0.05 mM MnCl2 (higher MnCl2 concentrations caused precipitation). |
Column 3: HiLoad 16/60 Superdex 200 gel filtration. |
Column 3 Buffer: 50 mM HEPES, pH 7.5; 250 mM NaCl. |
Column 3 Procedure: Dephosphorylated Pim1 protein was concentrated to 3ml and ran on a S200 gel filtration column collecting 1.8ml fractions. 10 mM DTT was added to the eluted protein for overnight storage. |
Column 3: Ion exchange Mono Q column |
Column 3 Buffer:
Buffer A:
50 mM HEPES, pH 7.5.
Buffer B: 50 mM HEPES, pH 7.5; 1000 mM NaCl. |
Column 3 Procedure: Pim1 was applied to Mono Q in buffer A and eluted from the column by linear gradient. |
Protein concentration: Pim1 fractions containing dephosphorylated protein were pooled and concentrated in Centricons (10kDa cut off). Phosphorylation state was confirmed using LC- ESI MS-Tof. |
Mass spectrometry characterization: The purified protein was homogeneous and had an experimental mass of 35545Da as expected from its primary structure. Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. Proteins were desalted prior to mass spectrometry by rapid elution off a C3 column with a gradient of 5-95% acetonitrile in water with 0.1% formic acid. |
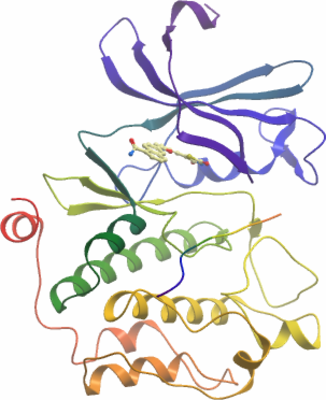
Crystallisation: The complex with compound A54 (6-{5-[(Z)-(2,4-dioxo-1,3-thiazolidin-5-ylidene)methyl]furan- 2-yl}-N-{3-[(4-ethylpiperazin-1-yl)methyl]- 5-(trifluoromethyl)phenyl}naphthalene-1-carboxamide) was grown by mixing 150nl of the protein (6mg/ml) with an equal volume of reservoir solution containing 0.2 M NaBr, 0.1 M BTProp pH 6.5, 20% PEG3350 and 10% EtGly and 5 mM solution of consensus peptide (ARKRRRHPSGPPTA-amide). Crystals grew to diffracting quality within a few days. Crystals were cryo-protected using the well solution supplemented with additional ethylene glycol and were flash frozen in liquid nitrogen. |
Data collection: Data were collected at a Rigaku FRE Superbright equipped with an RAXIS IV detector at 1.5Å. |