Entry Clone Source: MGC |
Entry Clone Accession: IMAGE:5114635 |
SGC Construct ID: CLK3A-c005 |
GenBank GI number: gi|4502885 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
CATATGCACCATCATCATCATCATTC
TTCTGGTGTAGATCTGGGTACCGAGA
ACCTGTACTTCCAATCCATGCAGAGC
AGTAAGCGCAGCAGCCGGAGTGTGGA
AGATGACAAGGAGGGTCACCTGGTGT
GCCGGATCGGCGATTGGCTCCAAGAG
CGATATGAGATTGTGGGGAACCTGGG
TGAAGGCACCTTTGGCAAGGTGGTGG
AGTGCTTGGACCATGCCAGAGGGAAG
TCTCAGGTTGCCCTGAAGATCATCCG
CAACGTGGGCAAGTACCGGGAGGCTG
CCCGGCTAGAAATCAACGTGCTCAAA
AAAATCAAGGAGAAGGACAAAGAAAA
CAAGTTCCTGTGTGTCTTGATGTCTG
ACTGGTTCAACTTCCACGGTCACATG
TGCATCGCCTTTGAGCTCCTGGGCAA
GAACACCTTTGAGTTCCTGAAGGAGA
ATAACTTCCAGCCTTACCCCCTACCA
CATGTCCGGCACATGGCCTACCAGCT
CTGCCACGCCCTTAGATTTCTGCATG
AGAATCAGCTGACCCATACAGACTTG
AAACCAGAGAACATCCTGTTTGTGAA
TTCTGAGTTTGAAACCCTCTACAATG
AGCACAAGAGCTGTGAGGAGAAGTCA
GTGAAGAACACCAGCATCCGAGTGGC
TGACTTTGGCAGTGCCACATTTGACC
ATGAGCACCACACCACCATTGTGGCC
ACCCGTCACTATCGCCCGCCTGAGGT
GATCCTTGAGCTGGGCTGGGCACAGC
CCTGTGACGTCTGGAGCATTGGCTGC
ATTCTCTTTGAGTACTACCGGGGCTT
CACACTCTTCCAGACCCACGAAAACC
GAGAGCACCTGGTGATGATGGAGAAG
ATCCTAGGGCCCATCCCATCACACAT
GATCCACCGTACCAGGAAGCAGAAAT
ATTTCTACAAAGGGGGCCTAGTTTGG
GATGAGAACAGCTCTGACGGCCGGTA
TGTGAAGGAGAACTGCAAACCTCTGA
AGAGTTACATGCTCCAAGACTCCCTG
GAGCACGTGCAGCTGTTTGACCTGAT
GAGGAGGATGTTAGAATTTGACCCTG
CCCAGCGCATCACACTGGCCGAGGCC
CTGCTGCACCCCTTCTTTGCTGGCTT
GACCCCTGAGGAGCGGTCCTTCCACA
CCTAAGACAGTAAAGGTGGATACGGA
TCCGAA
|
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfq^smQS
SKRSSRSVEDDKEGHLVCRIGDWLQE
RYEIVGNLGEGTFGKVVECLDHARGK
SQVALKIIRNVGKYREAARLEINVLK
KIKEKDKENKFLCVLMSDWFNFHGHM
CIAFELLGKNTFEFLKENNFQPYPLP
HVRHMAYQLCHALRFLHENQLTHTDL
KPENILFVNSEFETLYNEHKSCEEKS
VKNTSIRVADFGSATFDHEHHTTIVA
TRHYRPPEVILELGWAQPCDVWSIGC
ILFEYYRGFTLFQTHENREHLVMMEK
ILGPIPSHMIHRTRKQKYFYKGGLVW
DENSSDGRYVKENCKPLKSYMLQDSL
EHVQLFDLMRRMLEFDPAQRITLAEA
LLHPFFAGLTPEERSFHT
^ TEV cleavage site |
Tags and additions: Cleavable N-terminal His6 tag. |
Host: BL21 (DE3)R3-pRARE2 (Phage resistant strain).
|
Growth medium, induction protocol: 1ml from a 10 ml overnight culture containing 50µg/ml kanamycin were used to inoculate 1L cultures of LB containing 50µg/ml kanamycin. Cultures were grown at 37°C until the OD600 reached ~0.3 then the temperature was adjusted to 18°C. Expression was induced for 4 hours using 1 mM IPTG at an OD600 of 0.6. The cells were collected by centrifugation, resuspended in 30ml binding buffer, and frozen. The protein was co-expressed with λ-phoshatase.
Lysis buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% Glycerol; 50 mM L-Arg and L-Glu.
Extraction buffer, extraction method: The frozen cells were thawed on ice and binding buffer (plus 1 mM PMSF) were added to a final volume of 50ml. Cells were lysed using a high pressure cell disruptor. The lysate was centrifuged at 18,500prm for 50 minutes and the supernatant collected for purification. |
Column 1: Ni-affinity |
Column 1 Buffers:
Binding buffer:
50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 5 mM Imidazole; 50 mM L-Arg and L-Glu.
Wash buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 30 mM Imidazole; 50 mM L-Arg and L-Glu.
Elution buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 50 to 250 mM Imidazole; 50 mM L-Arg and L-Glu. |
Column 1 Procedure: In the cold-room, supernatant from the spun-down cell lysate was added to the DE-52 column, the flow through of which was passed onto the nickel resin column. After the lysate had passed through both columns, the columns were washed with approximately 150ml of binding buffer followed by approximately 150ml wash buffer. The flowthrough from the wash was collected and saved. A step-elution was performed by passing buffers with increasing imidazole concentrations over the nickel column and collecting the eluate. 5ml of each buffers containing 50mM, 100mM, 150mM and 250mM imidazole were used. Fractions were collected in 15ml falcon tubes. |
Column 2: Size Exclusion Chromatography. Superdex S75 16/60 HiLoad |
Column 2 Buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 50 mM L-Arg and L-Glu. |
Column 2 Procedure: The protein was concentrated and applied to an S75 16/60 Hi-Load gel filtration column equilibrated in 50 mM HEPES pH 7.5, 500 mM NaCl, 50 mM L-Arg and L-Glu using an ÄKTA Prime system. |
Mass spectrometry characterization: LC- ESI -MS TOF gave a measured mass of 44716.2Da for this construct as predicted from the sequence of this protein. |
Protein concentration: Protein was concentrated to 11.0mg/ml using an Amicon 10kDa cut-off concentrator. |
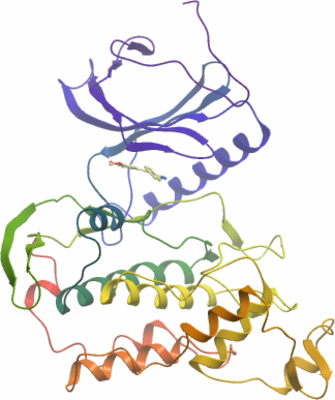
Crystallisation: Crystals grown at 4°C in 150nl sitting drops from a 1:1 ratio of protein to reservoir solution containing 0.1 M BTPropane pH 7.5, 20% PEG3350, 10% EtGly. L41 was co-crystallized with CLK3 by adding the compounds from a 50 mM DMSO stock to the concentrated protein (1 mM end concentration) . |
Data collection: Crystals were cryo-protected using the well solution supplemented by 20% ethylene glycol and flash frozen in liquid nitrogen.
X-ray source: Diffraction data were collected from a single crystal at the Swiss Light Source beamline PX10 at a single wavelength of 0.97924Å and the structure was refined to 2.09Å.
Phasing: The structure was solved by molecular replacement using an ensemble of known CLK1/3 structures as a starting model. |