Entry Clone Source: MGC |
Entry Clone Accession: BC032227 |
SGC Construct ID: ZFYVE16A-c008 |
GenBank GI number: gi|157426864 |
Vector: pNIC-Zb. |
Amplified construct sequence:
TACTTCCAATCCATGGAAGGCTTGGT
TTTGGGCCAGAAACAGCCTACTTGGG
TTCCTGATTCAGAAGCTCCAAACTGT
ATGAACTGCCAAGTCAAATTTACTTT
TACCAAACGGCGACACCATTGCCGAG
CATGTGGGAAAGTATTTTGTGGTGTC
TGTTGTAATAGGAAGTGTAAACTGCA
ATATCTAGAAAAGGAAGCAAGAGTAT
GTGTAGTCTGCTATGAAACTATTAGT
AAAGCTCAGGCATTTGAAAGGATGAT
GAGTCCAACTGGTTCTAATCTTAAGT
CTAATCATTCTGATGAATGTACTACT
GTCCAGCCTCCTCAGGAGAACCAAAC
ATCCAGTATACCTTCACCAGCAACTT
TGCCAGTCTCAGCACTTAAACAACCA
GGTGTTGAAGGACTATGACAGTAAAG
GTGGATA
|
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgvdnkfnkerrrarrei
rhlpnlnreqrrafirslrddpsqsa
nllaeakklndaqpkgtenlyfq^sm
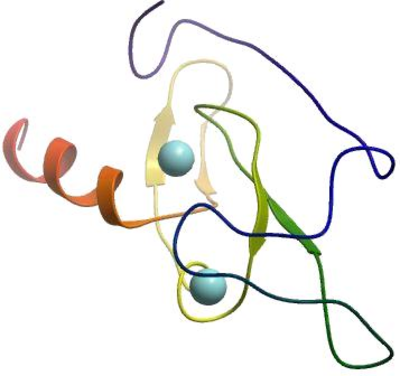
EGLVLGQKQPTWVPDSEAPNCMNCQV
KFTFTKRRHHCRACGKVFCGVCCNRK
CKLQYLEKEARVCVVCYETISKAQAF
ER*MMSPTGSNLKSNHSDECTTVQPP
QENQTSSIPSPATLPVSALKQPGVEG
L
^ TEV cleavage site
* Limited tryptic digestion is thought to have cleaved the protein here for crystallisation |
Tags and additions: N-terminal hexahistidine tag and Z tag cleavable by TEV protease (^). |
Host: BL21 (DE3)R3-pRARE2 (Phage resistant strain).
|
Growth medium, induction protocol: 50 mL of LB was inoculated with 1µl of glycerol stock and incubated overnight at 37°C while shaking in the presence of 50µg/ml kanamycin and 34µg/ml chloramphenicol. 1L of LB was inoculated with 10mL of overnight culture and grown while shaking at 37°C in the presence of antibiotic. Cultures were induced with 0.5 mM IPTG at OD600 = 0.9 and grown overnight at 21°C before harvesting by centrifugation.
Lysis buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% Glycerol; 0.5 mM TCEP.
Extraction buffer, extraction method: The frozen cells were thawed and lysed in lysis buffer by passage 4-5x through an Emulsiflex C5 homogenizer. The DNA was precipitated using 0.15% PEI (polyethyleneimine) pH 8. The cell lysate was spun down by centrifugation at 21.5K rpm at 4°C for 1h. The supernatant was recovered for purification. |
Column 1: Cation-exchange using a 5ml Hi Trap HP SP column (Amersham). The column was pre-equilibrated with binding buffer. |
Column 1 Buffers:
Binding buffer
: 50 mM HEPES, pH 7.5; 50 mM NaCl; 0.5 mM TCEP.
Elution buffer
: 50 mM HEPES, pH 7.5; 1 M NaCl; 0.5 mM TCEP. |
Column 1 Procedure: The supernatant was diluted by 50% with 50 mM HEPES pH 7.5 to reduce the ionic strength sufficiently to bind to the resin. The supernatant was applied onto the column using an ÄKTA purifier, which was then washed with 25mL binding buffer. ZFYVE16 protein was then eluted by applying a gradient of Elution buffer from 50 mM to 1 M NaCl at 1ml/min for 1h. 1.5mL fractions were collected across the entire gradient. |
Column 2: Size Exclusion Chromatography - S75 HiLoad 16/60 Superdex run on ÄKTA-Express. |
Gel Filtration Buffer: 50 mM HEPES, pH 7.5; 300 mM NaCl; 0.5 mM TCEP. |
Column 2 Procedure: Prior to applying the protein, the S75 16/60 column was washed and equilibrated with gel filtration buffer. The protein was concentrated to 3ml using an Amicon Ultra-15 filter with a 10kDa cut-off. The concentrated protein was directly applied onto the equilibrated S75 16/60 column, and run at a flow-rate of 1ml/min. Fractions containing the protein were pooled together. |
Enzymatic treatment: The tag was cleaved with TEV after Column 1 and the tag removed during gel filtration. |
Mass spectrometry characterization: The purified protein was homogeneous and had an experimental mass of 14596.7Da. The theoretical expected mass from the construct primary sequence is 14596.9Da. Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. Proteins were desalted prior to mass spectrometry by rapid elution off a C3 column with a gradient of 5-95% methanol in water with 0.1% formic acid. |
Protein concentration: Protein was concentrated to 15mg/ml using an Amicon 10kDa cut-off concentrator. |
Crystallisation: Protein was used at 8.9mg/ml in gel filtration buffer. Trypsin was added to the final sample at a concentration of 10µg/mL for limited in situ proteolysis during crystallisation. Crystals were grown at 4°C in 150nl sitting drops mixing 50nl protein solution with 100nl of a reservoir solution containing 20 mM NaNO3, 20% PEG 3350, 10% Ethylene Glycol. On mounting crystals were cryo-protected with an additional 20% ethylene glycol. |
Data collection: 1.1Å resolution.
X-ray source: Diamond Light Source beamline I04, λ = 0.9611Å. |