Entry Clone Source: Synthetic genes (codon-optimized for E. coli) |
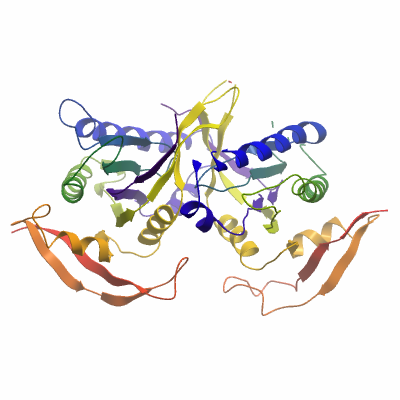
SGC Construct: This is a complex of two proteins; GUCY1A3 (aa 468 â 680) and a double mutant of GUCY1B3 (aa 408 â 610; G476C, C541S). |
SGC Construct 1 ID: GUCY1A3A-c060 |
Vector: pNH-TrxT Details [PDF]; Sequence [ FASTA] or [GenBank]
| Amplified DNA sequence:
ATGCACCATCATCATCATCATTCTTC
TGGTATGAGCGATAAAATTATTCACC
TGACTGACGACAGTTTTGACACGGAT
GTACTCAAAGCGGACGGGGCGATCCT
CGTCGATTTCTGGGCAGAGTGGTGCG
GTCCGTGCAAAATGATCGCCCCGATT
CTGGATGAAATCGCTGACGAATATCA
GGGCAAACTGACCGTTGCAAAACTGA
ACATCGATCAAAACCCTGGCACTGCG
CCGAAATATGGCATCCGTGGTATCCC
GACTCTGCTGCTGTTCAAAAACGGTG
AAGTGGCGGCAACCAAAGTGGGCGCA
CTGTCTAAAGGTCAGTTGAAAGAGTT
CCTCGACGCTAACCTGGCCGGTACCG
AGAACTTGTACTTCCAATCCATGCAA
GGTCAGGTCGTTCAAGCTAAGAAATT
CAGCAATGTGACGATGCTGTTTAGTG
ATATCGTAGGGTTCACCGCCATTTGC
TCACAGTGTTCCCCGCTGCAGGTCAT
CACTATGCTGAACGCACTGTACACAC
GCTTTGACCAACAGTGCGGCGAACTG
GATGTTTATAAAGTGGAGACAATTGG
TGACGCGTACTGTGTAGCTGGCGGTC
TGCATAAGGAAAGCGATACCCACGCC
GTCCAAATCGCACTGATGGCGCTGAA
AATGATGGAACTGTCGGATGAGGTTA
TGTCTCCGCATGGGGAACCGATTAAA
ATGCGTATTGGCCTGCATAGTGGTTC
AGTGTTCGCCGGGGTAGTCGGCGTTA
AGATGCCGCGCTATTGCCTGTTTGGT
AATAACGTGACTCTGGCAAATAAATT
TGAAAGCTGTTCCGTACCGCGTAAAA
TCAACGTATCGCCGACGACCTACCGC
CTGCTGAAGGACTGCCCGGGCTTCGT
TTTTACACCGCGTTCTCGCGAGGAAC
TGCCGCCGAATTTCCCGAGTGAGATT
CCGGGTATCTGTCACTTTCTGGATGC
GTATCAGCAGGGGACCAACTCAAAAC
CGTGCTTCCAAAAAAAGGACGTGGAA
GATGGCAATGCTAACTTTCTGGGTAA
AGCCAGCGGCATTGATTGA
|
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgmsdkiihltddsfdtd
vlkadgailvdfwaewcgpckmiapi
ldeiadeyqgkltvaklnidqnpgta
pkygirgiptlllfkngevaatkvga
lskgqlkefldanlagtenlyfq^SM
QGQVVQAKKFSNVTMLFSDIVGFTAI
CSQCSPLQVITMLNALYTRFDQQCGE
LDVYKVETIGDAYCVAGGLHKESDTH
AVQIALMALKMMELSDEVMSPHGEPI
KMRIGLHSGSVFAGVVGVKMPRYCLF
GNNVTLANKFESCSVPRKINVSPTTY
RLLKDCPGFVFTPRSREELPPNFPSE
IPGICHFLDAYQQGTNSKPCFQKKDV
EDGNANFLGKASGID
^ cleavage site |
Tags and additions: Cleavable N-terminal His6 âthioredoxin tag. |
SGC Construct 2 ID: GUCY1B3A-c313 |
Vector: pNIC-CTHF. Details [ PDF ]; Sequence [ FASTA ] or [ GenBank ]
|
Amplified DNA sequence:
ATGCACAAGCGTCCGGTTCCGGCCAA
ACGCTATGACAACGTGACTATTCTGT
TCTCCGGTATCGTAGGGTTTAATGCA
TTTTGTAGCAAACATGCGTCGGGCGA
GGGTGCTATGAAGATTGTTAACCTGC
TGAATGATCTGTACACACGTTTCGAT
ACGCTGACCGACTCTCGCAAAAACCC
GTTTGTGTATAAAGTCGAAACTGTTT
GCGATAAGTATATGACAGTGAGTGGT
CTGCCGGAGCCGTGCATCCATCACGC
CCGTTCAATTTGTCATCTGGCACTGG
ACATGATGGAAATTGCGGGGCAGGTA
CAGGTCGATGGCGAAAGCGTTCAAAT
CACGATTGGTATCCACACCGGGGAGG
TGGTAACTGGCGTCATTGGTCAGCGC
ATGCCGCGTTACTCCCTGTTCGGCAA
TACGGTTAACCTGACCTCCCGCACAG
AAACCACTGGTGAAAAAGGGAAAATC
AATGTGTCGGAGTATACATACCGTTG
TCTGATGTCTCCGGAAAACAGTGACC
CGCAATTTCATCTGGAGCACCGCGGC
CCGGTAAGCATGAAGGGTAAAAAAGA
ACCGATGCAGGTCTGGTTCCTGTCAC
GTAAGAATACGGGCACCGAAGAGACT
AAACAGGATGATGACGCAGAGAACCT
CTACTTCCAATCGCACCATCATCACC
ACCATGATTACAAGGATGACGACGAT
AAGTGA
|
Final protein sequence (Tag sequence in lowercase):
MHKRPVPAKRYDNVTILFSGIVGFNA
FCSKHASGEGAMKIVNLLNDLYTRFD
TLTDSRKNPFVYKVETVCDKYMTVSG
LPEPCIHHARSICHLALDMMEIAGQV
QVDGESVQITIGIHTGEVVTGVIGQR
MPRYSLFGNTVNLTSRTETTGEKGKI
NVSEYTYRCLMSPENSDPQFHLEHRG
PVSMKGKKEPMQVWFLSRKNTGTEET
KQDDDAENLYFQ^shhhhhhdykdddd
k
^ cleavage site |
Tags and additions: Cleavable C-terminal His6 âflag tag. |
Host: BL21(DE3)-R3-Chaperones (a BL21 derivative carrying the plasmid pGro7 from Takara, expressing the chaperones GroEL-GroES under arabinose regulation)
|
Growth medium, induction protocol: The expression plasmid was transformed into the host strain and plated on LB-agar containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol. Several colonies were combined to inoculate a 1ml culture in TB (+ 50 µg/ml kanamycin, 34 µg/ml chloramphenicol). The culture was grown overnight, glycerol was added to 15% v/v (from a 60% stock), and the resulting glycerol stock was frozen at -80°C in 100 µl aliquots. A loopful of cells from the glycerol stock was inoculated into 20-ml of TB medium containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol and grown overnight at 37°C. 2x 1L TB medium (containing 50 µg/ml kanamycin) were each inoculated with 10 ml of the overnight culture and grown in 2.5L UltraYield baffled flasks until OD600 of 1.0. Chaperone induction was performed by addition of 10mL 20% (w/v) Arabinose. Cells were cooled to 18°C, IPTG added to 0.1mM and growth continued at 18°C overnight. The cells were collected by centrifugation then the pellets were scraped out and transferred to 50-ml Falcon tubes and frozen at -80°C.
Lysis buffer: 50 mM Na-phosphate, pH 7.5, 500 mM NaCl, 30 mM imidazole, 1x Protease Inhibitors Cocktail Set VII (Calbiochem, 1/1000 dilution), Benzonase (3 µl per 25 ml, 30 units/ml).
PEI (polyethyleneimine) stock: 5% (w/v) pH 7.5, made from 50% solution (sigma P3143), pH adjusted with HCl.
Extraction method: Frozen cell pellets of both constructs were thawed briefly in a bath of warm water (20 - 37°C), mixed then transferred to ice. Lysis buffer was added, 2 x w/v. The cells were resuspended by agitating and disrupted by sonication. Nucleic acids and cell debris were removed by adding 0.15% PEI (polyethyleneimine) from a 5% (w/v, pH 7.5) stock, followed by centrifugation for 1 hour at 25,000 xg |
Column 1: Histrap FF 5ml (GE Healthcare). |
Column 1 Buffers:
Affinity buffer: 50 mM Na-phosphate, pH 7.5, 500 mM NaCl, 30 mM imidazole
Elution buffer: 50 mM Na-phosphate, pH 7.5, 500 mM NaCl, 300 mM imidazole |
Column 1 Procedure: The cell extract was loaded onto the column at 5 ml/minute on an ÄKTA-express system (GE Healthcare). The column was then washed with 10 volumes of loading buffer, 10 volumes of wash buffer, then eluted with elution buffer at 4 ml/min. The eluted peak of A280 was automatically collected. |
TEV cleavage and re-purification: The eluted GUCY complex protein was treated with TEV protease (His-tagged) overnight at 4°C, while being dialysed in 40mM Tris pH 8.0, 250 mM KCl, 30 mM imidazole, 1 mM TCEP, 10% glycerol, using 3000 MWCO dialysis tubing. The protease and other impurities were removed by passing through a 5mL NiNTA column. |
Column 2: Superdex 200 HiLoad 16/60 Gel Filtration 120ml |
Column 2 Buffers:
GF Buffer: 50 mM HEPES, pH 7.5, 300 mM NaCl, 5% glycerol. |
Column 2 Procedure: The eluted fraction was loaded and fractionated on the gel filtration column in GF buffer at 1.2 ml/min. 1.8ml fractions were collected at the A280 peaks. The fractions were analysed by SDS-PAGE and relevant fractions were pooled. |
Column 3: HiTrap-Q Sepharose XL 5 ml (GE Healthcare) |
Column 3 Buffers:
Buffer A: 25 mM Tris pH 8.5, 20 mM NaCl.
Buffer B: 25 mM Tris pH 8.5, 1000 mM NaCl. |
Column 3 Procedure: The column was prepared by pumping 10 column volumes of buffer B, followed by 25 column volumes of Buffer A. The protein sample was diluted in 25 mM Tris, pH 8.5, to a NaCl concentration corresponding with Buffer A. The sample was loaded on to the column at 4 mL/min. The protein was eluted using a gradient of Buffer B over 40 column volumes. A major peak was joined by a distinct 'shoulder'. The shoulder appeared to contain only GUCY1A3, whereas the major peak contained both GUCY1A3 and GUCY1B3. The major peak was pooled, concentrated to 16 mg/mL (estimated by A280). |
Protein concentration: The protein was then concentrated to 14.5 mg/ml (concentrations estimated by A280). |
Crystallisation: Crystals were grown by vapour diffusion at 20°C in 150 nl sitting drops. The drops were prepared by mixing 7 5nl of protein solution and 75 nl of precipitant consisting of 0.05M KH2PO4, 20% PEG 8000. The crystals were cryo-protected using the well solution supplemented with 25% ethylene glycol and flash-frozen in liquid nitrogen. |
Data collection:
X-ray source: Diamond I04
Resolution: 2.1Å
Diffraction data were collected from a single crystal at the Diamond synchrotron beamline I04 at a wavelength of 0.968627. The protein crystallised in space group P21, with two heterodimeric dimers in the asymmetric unit. The data was phased and an initial model was built using the BALBES server. Iterative rounds of manual building using COOT followed by refinement in using BUSTER was performed. The deposited structure was refined to a final resolution of 2.1A, R=0.168, Rfree=0.208, and assigned the PDB code 3UVJ. |