The ankyrin repeat domain of ANKRA2 is homologous (62% sequence identity) to
that of regulatory factor X-associated ankyrin-containing protein (RFXANK, also known
as ANKRA1 and RFX-B). Defects in RFXANK cause a rare immune disorder, bare
lymphocyte syndrome (BLS). RFXANK forms a trimeric complex with two other BLS-
associated proteins, regulatory factor X, 5 (RFX5) and RFXAP, to specifically recognize
the X-box regulatory element present at the enhancer region of genes that encode major
histocompatibility complex (MHC) proteins.
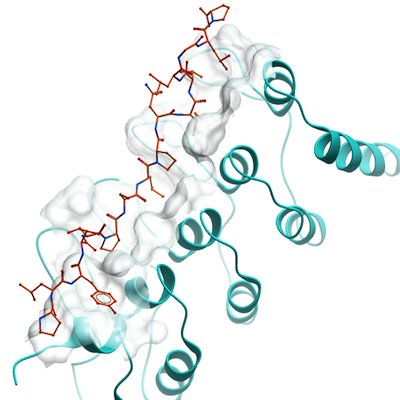
The structure of the complex of RFXANK (residues 90 to 260) bound to the RFX5
peptide (residues 167 to 183) indicates that RFX5 adopts a binding mode similar to that
found in the ANKRA2-HDAC4 complex. Superposition of the ANKRA2-HDAC4 and
RFXANK-RFX5 structures revealed that the three hydrophobic pockets of RFXANK
are occupied by Met172, Leu175, and Leu178 of RFX5. It was surprising thatMet172,
instead of Pro173, fits into the first pocket of RFXANK. Sequence analysis of the RFX5
peptide revealed that it contains another proline residue at the second position of the
PxLPxL motif. Proline has restrictions in ϕ-y space that arise from its five membered
ring, and this rigidity in the PP dipeptide causes the preceding methionine residue
(Met172) to occupy the first binding pocket. Met172 is not an ideal residue for the first
binding pocket because of its longer side chain, and this unfavorable arrangement may
compromise the overall binding affinity between RFXANK and RFX5. The resulting
complex structure confirmed that the His199 residue of mutant RFXANK R199H adopts
the same conformation as that of His257 in the ANKRA2 complexes and is involved in
binding to both Pro352 and Ile354 of HDAC4.