Ankyrin repeat family A protein 2 (ANKRA2) is an ankyrin repeat domain-
containing protein and was first identified as a binding partner for the cytoplasmic
domain of megalin, which is also known as low-density lipoprotein receptor-related
protein 2 (LRP2). Megalin is a multiligand-binding receptor found in the plasma
membrane of many absorptive epithelial cells, and it regulates the endocytosis of
various extracellular signaling proteins. ANKRA2 was identified as a binding partner of
histone deacetylase 4 (HDAC4) and HDAC5. HDAC4 and HDAC5 are members of the
class IIa subgroup of the HDAC superfamily and mainly function as co-repressors for
DNA binding transcription factors, such as myocyte enhancer factor 2 (MEF2) family
members, to repress gene expression in diverse developmental programs.
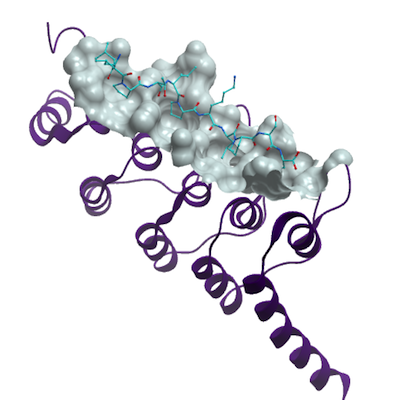
The crystal structure of the apo form of ANKRA2 shows that ANKRA2 contains
five ankyrin repeats at its C terminus, which are designated ANK1 to ANK5. The five
ankyrin repeats stack together, and the four hairpin loops that connect neighboring
ankyrin repeats are not only well aligned but also uniformly spaced apart from each
other. This architecture is characteristic of ankyrin repeat proteins. The structure of
ANKRA2 in complex with theHDAC4 peptide showed that the latter is bound in an
extended conformation with the same N terminus to C terminus orientation as is shown
by the binding groove defined by the inner a helices and inter repeat hairpin loops from
the five ankyrin modules. The structure of ANKRA2 in the complex is almost identical to
that of the unbound ANKRA2 with a root mean square deviation (RMSD) of 0.35 Å over
all of the aligned Ca atoms. The most notable feature of this structure is that each of the
middle three ankyrin repeats of ANKRA2 recognizes a residue from the HDAC4 peptide,
namely, three hydrophobic pockets are formed above the inner a helix of the middle three
ankyrin repeats of ANKRA2 and they accommodate the residues Pro349 (P349), Leu351
(L351), and Ile354 (I354) of the HDAC4 peptide.