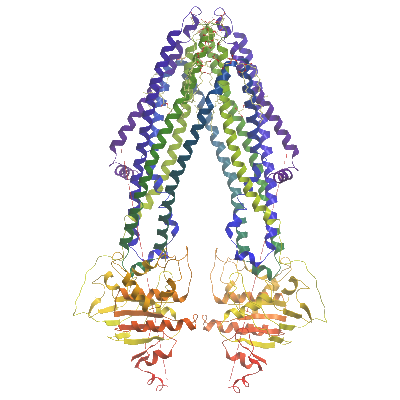
ABCB10 is a human mitochondrial ATP-binding cassette (ABC) transporter which is highly overexpressed in erythroid cells, cells that are committed to becoming red blood cells [1], [2], [3]. ABCB10 is embedded in the inner membrane of mitochondria, and is thought to pump its substrate out of the mitochondrial matrix into the intermembrane space. Human mitochondria have four ABC transporters, whereas yeast cells have three, including MDL1, which is highly homologous to ABCB10. Over expression of MDL1 in yeast cells lacking the gene for another mitochondrial ABC transporter, atm1, rescued cells under conditions of oxidative stress [4]. In mammals ABCB10 is important for hemoglobin and red blood cell formation and for protection of mitochondria against oxidative stress[5], [6]. Several alternative functional roles have been suggested for ABCB10 and its homologue MDL1 including transport of heme biosynthesis precursors [7], stabilisation of the iron transporter SLC25A37 [8] and peptide transport [9], [10]. However to date no substrate has been confirmed biochemically and the debate on ABCB10 function continues [3].
Here we present the structure of ABCB10 its nucleotide-free, apo state to 2.85Å resolution. This structure is similar to our previously deposited ABCB10 structures crystallised in the presence of DDM/cardiolipin. Here, ABCB10 exhibits an open inwards conformation with the nucleotide binding sites unoccupied and the residues preceding the Walker A motif adopting a helical conformation.