TP73A: P73 / Tumour Protein 73 Materials & Methods |
Entry Clone Source: NIH Mammalian Gene Collection |
Entry Clone Accession: BC117251 |
SGC Construct ID: TP73A-c009 |
GenBank GI number: |
Vector: pNIC-CTHF. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
TTAAGAAGGAGATATACTATGGCGCC
TGTCATCCCCTCCAACACCGACTACC
CCGGACCCCACCACTTTGAGGTCACT
TTCCAGCAGTCCAGCACGGCCAAGTC
AGCCACCTGGACGTACTCCCCGCTCT
TGAAGAAACTCTACTGCCAGATCGCC
AAGACATGCCCCATCCAGATCAAGGT
GTCCACCCCGCCACCCCCAGGCACTG
CCATCCGGGCCATGCCTGTTTACAAG
AAAGCGGAGCACGTGACCGACGTCGT
GAAACGCTGCCCCAACCACGAGCTCG
GGAGGGACTTCAACGAAGGACAGTCT
GCTCCAGCCAGCCACCTCATCCGCGT
GGAAGGCAATAATCTCTCGCAGTATG
TGGATGACCCTGTCACCGGCAGGCAG
AGCGTCGTGGTGCCCTATGAGCCACC
ACAGGTGGGGACGGAATTCACCACCA
TCCTGTACAACTTCATGTGTAACAGC
AGCTGTGTAGGGGGCATGAACCGGCG
GCCCATCCTCATCATCATCACCCTGG
AGATGCGGGATGGGCAGGTGCTGGGC
CGCCGGTCCTTTGAGGGCCGCATCTG
CGCCTGTCCTGGCCGCGACCGAAAAG
CTGATGAGGACCACTACCGGGAGGCA
GAGAACCTCTACTTCCAATC
|
Final protein sequence (Tag sequence in lowercase):
MAPVIPSNTDYPGPHHFEVTFQQSST
AKSATWTYSPLLKKLYCQIAKTCPIQ
IKVSTPPPPGTAIRAMPVYKKAEHVT
DVVKRCPNHELGRDFNEGQSAPASHL
IRVEGNNLSQYVDDPVTGRQSVVVPY
EPPQVGTEFTTILYNFMCNSSCVGGM
NRRPILIIITLEMRDGQVLGRRSFEG
RICACPGRDRKADEDHYREaenlyfq
^shhhhhhdykddddk
^ TEV cleavage site |
Tags and additions: TEV-cleavable C-terminal hexahistidine and FLAG tag |
Host: BL21(DE3)-R3-pRARE2 |
Growth Medium & Expression: A glycerol stock was used to inoculate a 10ml starter culture containing LB media with 50µg/ml Kanamycin and 34 µg/ml chloramphenicol. The starter culture was grown overnight at 37°C with shaking at 200 rpm. The following morning, two flasks containing 1 L LB/kanamycin/chloramphenicol were each inoculated with 5 ml of the starter culture. Cultures were incubated at 37°C with shaking at 180 rpm until an OD600nm ⥠0.6 was reached. The flasks were then cooled down to 18°C. Protein expression was induced by addition of IPTG to a final concentration of 0.2mM and expression carried out overnight. |
Cell harvest: Cells were harvested by centrifugation at 6000 rpm at 4°C for 15 min. |
Cell Lysis: Cell pellets from each flask were resuspended in 30ml binding buffer (50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole), transferred to 50 ml tubes, and stored at -20°C.
Lysis buffer: 50 mM Hepes pH 7.4, 500 mM NaCl, 5% Glycerol, 10 mM Imidazole pH 7.4, 0.5 mM TCEP, 1 tablet per 50 ml protease inhibitor cocktail EDTA-free (Roche)
Extraction buffer, extraction method: The frozen cells were thawed and 0.5mM TCEP, 1mM PMSF added to the cell suspension. The cells were lysed by ultrasonication over 12 min with the sonicator pulsing ON for 4 sec and OFF for 8 sec. The cell lysate was spun down by centrifugation at 20000 rpm at 4°C for 1 h. The supernatant was recovered for purification. |
Column 1: Anion-exchange for Nucleic acid removal with DEAE cellulose (DE52, Whatmann). 10 g of resin was suspended in 100 ml 2.5 M NaCl, and then applied onto a 2.5 x 20 cm column. The resin was then equilibrated with 100 ml binding buffer prior to loading the sample. |
Column 1 Buffers:
Binding buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole
Wash buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 30mM Imidazole
|
Column 1 Procedure: The supernatant was first applied onto the column by gravity flow, which was followed by a wash with 100 ml wash buffer. The column flow-through and wash was directly applied onto the following Ni-sepharose column |
Column 2: Ni-Affinity Chromatography 5ml of 50 % Ni-sepharose slurry (Amersham) was applied onto a 1.5 x 10 cm column. The column was first washed with deionised distilled H2O, and then equilibrated with binding buffer. |
Column 2 Buffers:
Binding buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole
Wash buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 30mM Imidazole
Elution buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 50 to 250mM Imidazole
|
Column 2 Procedure: The supernatant was applied by gravity flow onto the Ni-sepharose column. The column was washed with wash buffer to remove nonspecifically binding proteins. The bound target protein was eluted by applying a step gradient of imidazole (5 ml fractions of elution buffer supplemented with 50mM, 100mM, 150mM and 250mM imidazole). The protein content of collected fractions was visualized using SDS-PAGE and fractions containing sufficient TP73A (relative to contaminating proteins) were pooled. 10mM DTT was added to the pooled fractions for overnight storage at 4°C. |
Enzymatic Treatment:TEV protease cleavage. Pooled fractions were treated with TEV protease overnight at 4°C. |
Column 3: Size Exclusion Chromatography S200 HiLoad 16/60 Superdex run on Äaut;KTA-Express. |
Column 3 Buffers:
Gel filtration buffer: 300mM NaCl, 50mM HEPES pH 7.5, 0.5mM TCEP.
|
Column 3 Procedure: Prior to applying the protein, the S200 16/60 column was washed and equilibrated with gel filtration buffer. Eluted protein from the Ni-sepharose column was cleaved with TEV protease and concentrated to 1ml using an Amicon Ultra-15 filter with a 10kDa cut-off. The concentrated protein was directly applied onto the equilibrated S200 16/60 column, and run at a flow-rate of 1 ml/min. Fractions were visualized using SDS-PAGE and those containing TP73A were pooled.
|
Mass spectrometry characterization:
Observed mass: 23184.05 Da
Expected mass: 23313.6 Da
|
Mass spec procedure: The purified native protein was homogeneous and had an experimental mass after tag cleavage of 23184.05 consistent with loss of the N-terminal initiator methionine (calculated MW of this species = 23313.6). Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. |
ASPP2 / Apoptosis Stimulating p53 Protein 2 (aka TP53BP2A: Tumour Protein 53 Binding Protein 2) Materials & Methods |
Entry Clone Source: NIH Mammalian Gene Collection |
Entry Clone Accession: BC040247 |
SGC Construct ID: TP53BP2A-c003 |
GenBank GI number: |
Vector: pCOEX-LIC1 Details [PDF ]; Sequence [ FASTA ] |
Amplified construct sequence:
GAACGAAGTTCGCTTCTTCCTTCGTC
TACTTCCAATCCATGCCTGAAATCAC
CGGGCAGGTCTCTCTGCCTCCTGGTA
AAAGGACAAACTTGCGTAAAACTGGC
TCAGAGCGTATCGCTCATGGAATGAG
GGTGAAATTCAACCCCCTTGCTTTAC
TGCTAGATTCGTCTTTGGAGGGAGAA
TTTGACCTTGTACAGAGAATTATTTA
TGAGGTTGATGACCCAAGCCTCCCCA
ATGATGAAGGCATCACGGCTCTTCAC
AATGCTGTGTGTGCAGGCCACACAGA
AATCGTTAAGTTCCTGGTACAGTTTG
GTGTAAATGTAAATGCTGCTGATAGT
GATGGATGGACTCCATTACATTGTGC
TGCCTCATGTAACAACGTCCAAGTGT
GTAAGTTTTTGGTGGAGTCAGGAGCC
GCTGTGTTTGCCATGACCTACAGTGA
CATGCAGACTGCTGCAGATAAGTGCG
AGGAAATGGAGGAAGGCTACACTCAG
TGCTCCCAATTTCTTTATGGAGTTCA
GGAGAAGATGGGCATAATGAATAAAG
GAGTCATTTATGCGCTTTGGGATTAT
GAACCTCAGAATGATGATGAGCTGCC
CATGAAAGAAGGAGACTGCATGACAA
TCATCCACAGGGAAGACGAAGATGAA
ATCGAATGGTGGTGGGCGCGCCTTAA
TGATAAGGAGGGATATGTTCCACGTA
ACTTGCTGGGACTGTACCCAAGAATT
AAACCAAGACAAAGGAGCTTGGCCTG
ACAGTAAAGGTGGATA
|
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfq^smpE
ITGQVSLPPGKRTNLRKTGSERIAHG
MRVKFNPLALLLDSSLEGEFDLVQRI
IYEVDDPSLPNDEGITALHNAVCAGH
TEIVKFLVQFGVNVNAADSDGWTPLH
CAASCNNVQVCKFLVESGAAVFAMTY
SDMQTAADKCEEMEEGYTQCSQFLYG
VQEKMGIMNKGVIYALWDYEPQNDDE
LPMKEGDCMTIIHREDEDEIEWWWAR
LNDKEGYVPRNLLGLYPRIKPRQRSL
A
^ TEV cleavage site |
Tags and additions: TEV-cleavable N-terminal His7 tag. |
Host: BL21(DE3)-R3-pRARE2 |
Growth Medium & Expression: A glycerol stock was used to inoculate a 10ml starter culture containing LB media with 34 µg/ml chloramphenicol. The starter culture was grown overnight at 37°C with shaking at 200 rpm. The following morning, six flasks containing 1 L LB/chloramphenicol were each inoculated with 5 ml of the starter culture. Cultures were incubated at 37°C with shaking at 180 rpm until an OD600nm ⥠0.6 was reached. The flasks were then cooled down to 18°C. Protein expression was induced by addition of IPTG to a final concentration of 0.2mM and expression carried out overnight. |
Cell harvest: Cells were harvested by centrifugation at 6000 rpm at 4°C for 15 min. |
Cell Lysis: Cell pellets from each flask were resuspended in 30ml binding buffer (50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole), transferred to 50 ml tubes, and stored at -20°C.
Extraction buffer, extraction method: The frozen cells were thawed and 0.5mM TCEP, 1mM PMSF added to the cell suspension. The cells were lysed using an Emulsiflex C3 homogeniser. The cell lysate was clarified by centrifugation at 20000 rpm at 4°C for 1 h. The supernatant was recovered for purification and filtered through a syringe filter with 1.2μm pore size. |
Column 1: Ni-Affinity Chromatography 2ml Ni-sepharose slurry applied to a 1.5 x 10 cm column |
Column 1 Buffers:
Binding buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole
Wash buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 30mM Imidazole
Elution buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 50 to 250mM Imidazole
|
Column 1 Procedure: The supernatant was first applied onto the column by gravity flow, which was followed by a wash with 100 ml wash buffer. The column flow-through and wash was directly applied onto the following Ni-sepharose column |
Enzymatic Treatment:TEV protease cleavage. Pooled fractions were treated with TEV protease overnight at 4°C. |
Column 2: Ion Exchange Chromatography HiTrap Q HP 5ml run on Akta Express |
Column 2 Buffers:
Buffer A: 50mM HEPES, pH 7.5; 50mM NaCl; 0.5mM TCEP
Buffer B: 50mM HEPES, pH 7.5; 1M NaCl; 0.5mM TCEP
Sample buffer: 50mM HEPES, pH 7.5; 0.5mM TCEP
|
Column 2 Procedure: Prior to applying the protein, the HiTrap Q HP 5ml column was washed with buffer B and equilibrated in buffer A. Eluted protein from the Ni-sepharose column was cleaved with TEV protease and diluted in sample buffer 10x to give a final buffer concentration of 50mM NaCl. The diluted protein was applied onto the equilibrated HiTrap Q column and the column washed with 4 column volumes of buffer A. Bound proteins were eluted with a gradient up to 50% buffer B over 100ml at 4 ml/minute and 1ml fractions were collected by peak detection. Fractions were visualized using SDS-PAGE and those containing ASPP2 were pooled. |
Column 3: Size Exclusion Chromatography S75 HiLoad 16/60 Superdex run on Äaut;KTA-Express. |
Column 3 Buffers:
Gel filtration buffer: 10mM HEPES pH 7.5, 150mM NaCl, 0.5mM TCEP
|
Column 3 Procedure: Prior to applying the protein, the S75 16/60 column was washed and equilibrated with gel filtration buffer. Eluted protein from the HiTrap Q HP column was concentrated to 1ml using an Amicon Ultra-15 filter with a 10kDa cut-off. The concentrated protein was directly applied onto the equilibrated S75 16/60 column, and run at a flow-rate of 1 ml/min. Fractions were visualized using SDS-PAGE and those containing ASPP2 were pooled. |
Mass spectrometry characterization:
Observed mass: 26924.43 Da
Expected mass: 26924.6 Da
|
Mass spec procedure: The purified native protein was homogeneous and had an experimental mass after tag cleavage of 26924.43 consistent with the calculated intact mass (calculated MW of this species = 26924.6). Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. |
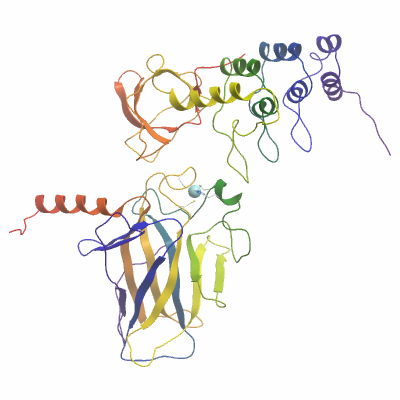
Complex materials & methods |
Concentration and complex formation: The two proteins were mixed in a 1:1 ratio, then buffer exchanged (into 10mM HEPES pH 7.5, 0.5mM TCEP) and concentrated to a final concentration of 11mg/ml (measured by OD280 based on extinction coefficient 58330) in an Amicon Ultra-15 filter with a 10 kDa cut-off. |
Crystallisation: The complete complex at 11mg/ml was buffered in 10mM HEPES, pH 7.5, 0.5 mM TCEP. Crystals were grown at 4°C in 150 nl sitting drops mixing 100 nl protein solution with 50 nl of a reservoir solution containing 1.0M sodium potassium phosphate and 0.1M acetate pH 4.5. On mounting crystals were cryo-protected with an additional 20% ethylene glycol. |
Data collection:
Resolution: 2.6Å.
X-ray source: Diamond Light Source beamline I03.
Crystals of TP73A diffracted to a resolution of 2.6 Å (scaled resolution). A full dataset was collected at 100 K on Diamond Light Source beamline I03. Crystals belonged to the space group I121 with unit-cell parameters a= 132.8 Å b= 170.1 Å c= 177.6 Å, α=90° β= 92° γ= 90°. Six complete complexes (12 chains in total) were present in the asymmetric unit. Data were indexed and integrated using iMOSFLM and scaled using SCALA. Phases were found using molecular replacement in PHASER using PDB entries 1YCS (chain B) and 2XWC as search models. The structure was initially refined and modified using alternate rounds of REFMAC5 and COOT, and completed using BUSTER |