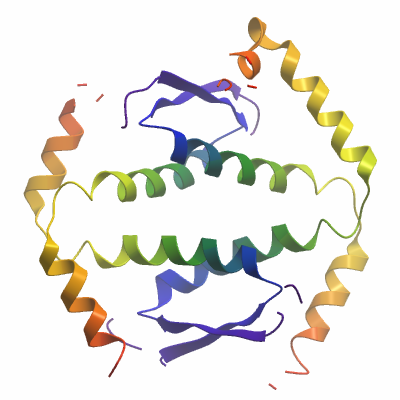
Human p63 Tetramerization Domain (TD)
PDB code: 4A9Z
p63 (TP63; TP73L, Tumor protein p73-like) belongs with p53 and p73 to the p53 family of proteins. All p53 family members exhibit high amino acid identity, especially among their transactivation (TAD), DNA-binding (DBD), and tetramerization domain (TD). Two different promoters in the p63 gene give rise to isoforms either containing an N-terminal transactivation domain (TA) or lacking this domain (DeltaN). In addition, alternative splicing produces several C-terminal splice variants. Whereas the isoforms with the shortest C termini (TAp63γ,δ,ε and TAp73γ,δ) show a very similar domain organization compared to p53, the isoforms with the extended C-termini (TAp63α and TAp73α) contain additional domains that are not present in p53, namely a sterile alpha motif (SAM) domain and a transcription inhibition domain (TID). The different isoforms can act as transcriptional activators or repressors depending of the isoform. p63 is thereby able to utilize p53 DNA binding sites but unique p63 consensus recognition sites have also been identified.
Knockout studies have shown that p63 plays a key role in the development of limbs, epithelial cells and cranium as those mice display severe deformations of limbs as well as of epithelia including skin, breast, urothelia, and prostate. p63-/- mice lack mammary glands, hair follicles, and teeth. Due to the slightly different phenotypes of the knockout models created it is under discussion if p63 is required for stem cell maintenance or commitment to stratification. Likewise the role of p63 in tumour formation is still unclear as in one p63+/- model mice developed spontaneous tumours whereas in the other they did not. Likewise p63 has been found to be over-expressed in a number of tumours but also been found to be reduced in others implicating that it has the potential to function as a tumour suppressor or as a proto-oncogene. Additionally p63 plays in conjunction with p73 a role in apoptosis and is essential in a process of DNA damage-induced oocyte death.
Mutations in p63 can lead to a variety of human human syndromes involving limb and ectodermal development including ADULT syndrome (acro-dermato-ungual- lacrimal-tooth syndrome), AEC syndrome (ankyloblepharon- ectodermal defects-cleft lip/palate), also known as Hay-Wells syndrome, EEC3, ectrodactyly, ectodermal dysplasia, and cleft lip/palate syndrome 3, SHFM4, split-hand/foot malformation 4, LMS, limb-mammary syndrome and RHS, Rapp-Hodgkin syndrome. In collaboration with the group of Volker Dötsch (Frankfurt University) we determined the crystal structure of the p63 tetramerization domain (TD), a domain that also forms hetero-tetramers with p73 but not with p53. Similar to structures of other family members, p63 forms a dimer of dimers with D2 symmetry. In contrast to p53, p63 and p73 contain an additional helix that stabilizes the tetrameric state. This second helix in the tetramerization domain seems to be an evolutionarily conserved structural element being present in many invertebrate p53 protein family members as well.