Entry Clone Source: Site-directed mutagenesis |
Entry Clone Accession: GI:125987600 |
SGC Construct ID: CDKL2A-c006 |
Vector: pFB-LIC-Bse
|
Amplified DNA sequence:
TACTTCCAATCCATGGAAAAATATGA
AAACCTGGGTTTGGTTGGAGAAGGGA
GTTATGGAATGGTGATGAAGTGTAGG
AATAAAGATACTGGAAGAATTGTGGC
CATAAAGAAGTTCTTAGAAAGTGACG
ATGACAAAATGGTTAAAAAGATTGCA
ATGCGAGAAATCAAGTTACTAAAGCA
ACTTAGGCATGAAAACTTGGTGAATC
TCTTGGAAGTGTGTAAGAAAAAAAAA
CGATGGTACCTAGTCTTTGAATTTGT
TGACCACACAATTCTTGATGACTTGG
AGCTCTTTCCAAATGGACTAGACTAC
CAAGTAGTTCAAAAGTATTTGTTTCA
GATTATTAATGGAATTGGATTTTGTC
ACAGTCACAATATCATACACAGAGAT
ATAAAGCCAGAGAATATATTAGTCTC
CCAGTCTGGCGTTGTCAAGCTATGCG
ATTTTGGATTTGCGCGAACATTGGCA
GCTCCTGGGGAGGTTTATGATGATGA
AGTGGCAACCCGATGGTACAGAGCTC
CAGAACTATTGGTTGGTGATGTCAAG
TATGGCAAGGCTGTTGATGTGTGGGC
CATTGGTTGTCTGGTAACTGAAATGT
TCATGGGGGAACCCCTATTTCCTGGA
GATTCTGATATTGATCAGCTATATCA
TATTATGATGTGTTTAGGTAATCTAA
TTCCAAGGCATCAGGAGCTTTTTAAT
AAAAATCCTGTGTTTGCTGGAGTAAG
GTTGCCTGAAATCAAGGAAAGAGAAC
CTCTTGAAAGACGCTATCCTAAGCTC
TCTGAAGTGGTGATAGATTTAGCAAA
GAAATGCTTACATATTGACCCCGACA
AAAGACCCTTCTGTGCTGAGCTCCTA
CACCATGATTTCTTTCAAATGGATGG
ATTTGCTGAGAGGTTTTCCCAAGAAC
TACAGTTAAAAGTACAGAAAGATGCC
TGACAGTAAAGGTGGATA
|
Final protein sequence (Tag sequence in lowercase):
mghhhhhhssgvdlgtenlyfq^sME
KYENLGLVGEGSYGMVMKCRNKDTGR
IVAIKKFLESDDDKMVKKIAMREIKL
LKQLRHENLVNLLEVCKKKKRWYLVF
EFVDHTILDDLELFPNGLDYQVVQKY
LFQIINGIGFCHSHNIIHRDIKPENI
LVSQSGVVKLCDFGFARTLAAPGEVY
DDEVATRWYRAPELLVGDVKYGKAVD
VWAIGCLVTEMFMGEPLFPGDSDIDQ
LYHIMMCLGNLIPRHQELFNKNPVFA
GVRLPEIKEREPLERRYPKLSEVVID
LAKKCLHIDPDKRPFCAELLHHDFFQ
MDGFAERFSQELQLKVQKDA
^ TEV cleavage site |
Tags and additions: Tev-cleavable (^) C-terminal hexahistidine tag. |
Host: SF9 Spodoptera frugiperda Insect cells
|
Growth medium, induction protocol: 2L of SF9 cells at a density of 2million/ml were infected with 10ml of Virus/L. Cells were incubated at 27°C in the shaker incubator and harvested after 48 hours. Cells were harvested by centrifugation at 900xG at 4°C for 15 min. Cell pellets from each flask (1l volume) were resuspended in 15ml binding buffer, transferred to 50ml tubes, and stored at -20°C.
Binding buffer50 mM HEPES, pH 7.5; 500 mM NaCl; 5 mM Imidazole; 5% Glycerol.
Extraction buffer, extraction method: The frozen cells were thawed and protease inhibitor SET V (Calbiochem) added to the cell suspension at 1:100 dilution. The cells were lysed by ultrasonication over 20 min with the sonicator pulsing ON for 5 sec and OFF for 10 sec. The cell lysate was spun down by centrifugation at 21,000 rpm at 4°C for 1 h. The clarified cell extract was filtered using syringe filters with a 1.2µm pore size. |
Column 1: Ni-Affinity Chromatography 2ml Ni-sepharose slurry applied to a 1.5 x 10 cm column |
Column 1 Buffers:
Binding buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 5 mM Imidazole.
Wash buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 30 mM Imidazole.
Elution buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol, 50 to 250 mM Imidazole. |
Column 1 Procedure: 2ml of 50% Ni-sepharose slurry (Amersham) was added to the filtered lysate, which was incubated with the Ni-sepharose for 1 hour at 4°C with slow rotation to maximize binding. The lysate was then applied to a 1.5 x 10 cm column by gravity flow. The remaining resin was then washed with 2x50ml binding buffer and 1x30ml wash buffer to remove nonspecifically binding proteins. The bound target protein was eluted by applying a step gradient of imidazole (5 ml fractions of elution buffer supplemented with 50mM, 100mM, 150mM and 250mM imidazole). The protein content of collected fractions was visualized using SDS-PAGE and fractions containing CDKL2A were pooled |
Column 2: Size Exclusion Chromatography S75 HiLoad 26/60 Superdex run on ÄKTAprime |
Column 2 Buffers:
Gel Filtration buffer: 10mM HEPES pH7.5, 500mM NaCl, 5% Glycerol, 1mM TCEP.
|
Column 2 Procedure: Prior to applying the protein, the S75 26/60 column was washed and equilibrated with gel filtration buffer. Eluted protein from Ni-sepharose column was concentrated to 2ml using an Amicon Ultra-15 filter with a 10kDa cut-off. The concentrated protein was directly applied onto the equilibrated S75 16/60 column, and run at a flow-rate of 2.5 ml/min. 3ml fractions were collected and visualized using SDS-PAGE. Those containing CDKL2A were pooled. |
Column 3: Ion Exchange Chromatography HiTrap Q HP 5ml run on Äkta Express |
Column 3 Buffers:
IEX Buffer A (Binding): 50mM HEPES pH7.5, 50mM NaCl, 5% Glycerol, 0.5mM TCEP.
IEX Buffer B (Elution):50mM HEPES pH7.5, 1M NaCl, 5% Glycerol, 0.5mM TCEP.
IEX Sample Buffer:50mM HEPES pH7.5, 1M NaCl, 5% Glycerol, 0.5mM TCEP.
|
Column 3 Procedure: Prior to applying the protein, the HiTrap Q HP 5ml column was washed with buffer B and equilibrated in buffer A. Eluted protein from the gel filtration column was diluted in sample buffer 10x to give a final buffer concentration of 50mM NaCl. The diluted protein was applied onto the equilibrated HiTrap Q column and the column washed with 4 column volumes of buffer A. Bound proteins were eluted with a gradient up to 50% buffer B over 100ml at 4 ml/minute and 1ml fractions were collected by peak detection. Fractions were visualized using SDS-PAGE and those containing CDKL2A were pooled. After concentration, some of the protein was immediately used for crystallization experiments and the rest was divided into 100µl aliquots and stored at -80°C. |
Protein concentration: The protein was concentrated to a final concentration of 10mg/ml (measured by OD280 based on extinction coefficient 32890) in an Amicon Ultra-15 filter with a 10 kDa cut-off. |
Mass spectrometry characterization: The purified protein was homogeneous and had an experimental mass of 38244.17 Da. The theoretical expected mass from the construct sequence is 38331.5 Da. The difference corresponds to loss of the N-terminal methionine and acetylation of the resulting N-terminus. Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. Proteins were desalted prior to mass spectrometry by rapid elution off a C3 column with a gradient of 5-95% methanol in water with 0.1% formic acid. |
Crystallisation: Protein at 10mg/ml was buffered in 10mM HEPES, pH 7.5, 500mM NaCl, 1 mM TCEP. 5-Amino-3-((4-(aminosulfonyl)phenyl)amino)-N-(2,6-difluorophenyl)-1H-1,2,4-triazole-1-carbothioamide was added to the final sample. Crystals were grown at 4°C in 150 nl sitting drops mixing 50 nl protein solution with 100 nl of a reservoir solution containing 0.2M sodium chloride, 0.25% PEG 3350 and 0.1M Tris pH 8.5. On mounting crystals were cryo-protected with an additional 25% ethylene glycol. |
Data collection:
X-ray source: Diamond I02
Resolution: 1.6Å
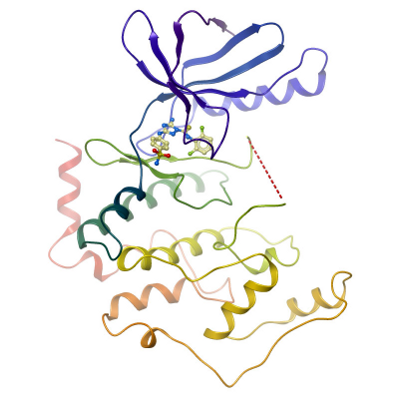
Crystals of CDKL2A diffracted to a resolution of 1.6 Å (scaled resolution). A full dataset was collected at 100 K on Diamond Light Source beamline I02. Crystals belonged to the Orthorhombic space group P212121 with unit-cell parameters a=67.8 Å b=70.2 Å c=83.6 Å, α=90° β= 92° γ= 90°. Only one molecule was present in the asymmetric unit. Data were indexed and integrated using iMOSFLM and scaled using SCALA. Phases were found using molecular replacement in PHASER. PHENIX.SCULPTOR was used to optimize the PDB entry 3NIZ for use as a search model. The structure was refined and modified using alternate rounds of REFMAC5 and COOT, and the final model validated using the JCSG Quality Control Server.
|