Entry Clone Source: Prof Nick La Thangue, University of Oxford |
Entry Clone Accession: N/A |
SGC Construct ID: TTC5MMA-c001 |
Vector: pET28a
|
Amplified DNA sequence:
ATGGGCAGCAGCCATCATCATCATCA
TCACAGCAGCGGCCTGGTGCCGCGCG
GCAGCCATATGGCTAGCATGACTGGT
GGACAGCAAATGGGTCGCGGATCCAT
GATGGCTGATGAAGAGGAAGAAGCGA
AGCACGTCTTGCAGAAATTGCAGGGA
CTGGTGGATCGGCTCTACTGTTTTCG
AGACAGTTACTTTGAGACACATAGTG
TCGAAGATGCAGGACGGAAGCAGCAG
GATGTACAGGAAGAGATGGAGAAGAC
CCTGCAGCAGATGGAGGAAGTACTCG
GTTCTGCCCAGGTTGAGGCACAGGCT
CTGATGCTGAAGGGGAAGGCACTGAA
TGTGACTCCTGATTATAGCCCTGAGG
CCGAGGTGCTTCTCTCCAAGGCCGTG
AAGCTGGAGCCTGAGCTGGTGGAAGC
CTGGAACCAGCTGGGTGAGGTGTACT
GGAAGAAAGGAGATGTCACATCTGCC
CACACCTGCTTCTCAGGAGCCCTCAC
CCACTGCAAGAACAAAGTCTCTCTGC
AGAACTTGTCCATGGTGCTCCGCCAG
CTGCAGACCGACTCTGGAGATGAACA
TTCTCGCCACGTCATGGACAGCGTCC
GGCAGGCTAAGTTGGCCGTGCAGATG
GATGTCCTTGATGGCCGCTCCTGGTA
TATCCTGGGGAATGCATATCTTTCTC
TTTATTTCAATACTGGCCAGAACCCT
AAGATCTCCCAGCAAGCCCTCAGTGC
CTATGCTCAAGCAGAGAAGGTGGACA
GGAAAGCATCTAGCAACCCTGACCTT
CATCTCAACAGGGCGACGTTACATAA
ATATGAGGAGAGTTATGGGGAGGCCC
TTGAGGGTTTCTCTCAGGCTGCAGCG
CTGGACCCTGCGTGGCCAGAGCCCCA
GCAACGAGAACAGCAACTCTTGGAAT
TCCTCAGTAGGCTAACCAGCCTCCTG
GAGAGCAAGGGGAAGACAAAGCCCAA
GAAGCTGCAGAGCATGCTGGGAAGCT
TGCGCCCAGCTCATCTGGGCCCCTGT
GGTGATGGGCGCTATCAGTCGGCCTC
TGGGCAGAAGATGACCCTGGAGCTTA
AGCCACTGAGCACCCTGCAGCCTGGT
GTGAACAGTGGCACCGTGGTCCTGGG
AAAGGTGGTGTTCAGCCTGACCACAG
AGGAGAAAGTCCCCTTCACGTTTGGC
TTGGTAGATTCGGATGGTCCCTGCTA
TGCAGTGATGGTGTATAATGTGGTGC
AGAGCTGGGGAGTGCTCATCGGGGAC
TCTGTAGCTATTCCTGAGCCCAACCT
TCGGCATCATCAAATCCGGCACAAGG
GAAAGGACTATTCCTTCTCCAGCGTG
CGTGTGGAAACGCCTCTTCTGCTGGT
GGTGAATGGAAAGCCACAGAACTCCA
GCAGTCAAGCCTCTGCCACAGTAGCT
TCAAGGCCACAGTGTGAATGA
|
Final protein sequence (Tag sequence in lowercase):
mgsshhhhhhssglvpr^gshmasmt
ggqqmgrgsMMADEEEEAKHVLQKLQ
GLVDRLYCFRDSYFETHSVEDAGRKQ
QDVQEEMEKTLQQMEEVLGSAQVEAQ
ALMLKGKALNVTPDYSPEAEVLLSKA
VKLEPELVEAWNQLGEVYWKKGDVTS
AHTCFSGALTHCKNKVSLQNLSMVLR
QLQTDSGDEHSRHVMDSVRQAKLAVQ
MDVLDGRSWYILGNAYLSLYFNTGQN
PKISQQALSAYAQAEKVDRKASSNPD
LHLNRATLHKYEESYGEALEGFSQAA
ALDPAWPEPQQREQQLLEFLSRLTSL
LESKGKTKPKKLQSMLGSLRPAHLGP
CGDGRYQSASGQKMTLELKPLSTLQP
GVNSGTVVLGKVVFSLTTEEKVPFTF
GLVDSDGPCYAVMVYNVVQSWGVLIG
DSVAIPEPNLRHHQIRHKGKDYSFSS
VRVETPLLLVVNGKPQNSSSQASATV
ASRPQCE
^ Thrombin cleavage site |
Tags and additions: N-terminal His6 T7 Tag . |
Host: BL21(DE3)-R3 (selected phage resistant strain)
|
Growth medium, induction protocol: A glycerol stock was used to inoculate a 50ml starter culture containing LB media with 50µg/ml Kanamycin. The starter culture was grown overnight at 37°C with shaking at 200 rpm. The following morning, four flasks containing 1 L LB/kanamycin were each inoculated with 7 ml of the starter culture. Cultures were incubated at 37°C with shaking at 160 rpm until an OD600nm = 0.5 was reached. The flasks were then cooled down to 18°C and 0.5mM IPTG added to induce protein expression overnight. Cells were harvested by centrifugation at 6000 rpm at 4°C for 20 min and stored at -20°C. Selenomethionine-labelled protein was expressed and purified similarly substituting Selenomethionine Expression Media (Molecular Dimensions) for Luria Broth (LB).
Extraction buffer, extraction method: 2L cell pellets were resuspended in 50ml binding buffer (50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole) supplemented with 1 mM TCEP and a 1mM PMSF. The cells were lysed by ultrasonication over 10 min with the sonicator pulsing ON for 2 sec and OFF for 4 sec. Insoluble material was removed by centrigugation at 16500 rpm for 1hr at 4°C. |
Column 1: Anion-exchange for Nucleic acid removal with DEAE cellulose (DE52, Whatmann)
10 g of resin was suspended in 100 ml 2.5 M NaCl, and then applied onto a 2.5 x 20 cm column. The resin was then equilibrated with 100 ml binding buffer prior to loading the sample.
|
Column 1 Buffers:
Binding buffer: 50 mM Hepes, pH 7.5; 500 mM NaCl; 5% Glycerol; 5 mM imidazole. |
Column 1 Procedure: The supernatant was first applied onto the column by gravity flow, which was followed by a wash with 50 ml binding buffer. The column flow-through and wash was directly applied onto a Ni-sepharose column. |
Column 2: Ni-Affinity Chromatography |
Column 2 Buffers:
Binding buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole.
Wash buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 30mM Imidazole.
Elution buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 50 to 250mM Imidazole.
|
Column 2 Procedure: The supernatant was applied by gravity flow onto the Ni-sepharose column. The column was then washed with 50 ml binding buffer and 30 ml wash buffer. The bound protein was eluted by applying a step gradient of imidazole (5 ml fractions of elution buffer sequentially containing 50mM, 100mM, 150mM or 250mM imidazole). 10mM DTT was added to each fraction collected for overnight storage at 4°C. |
Enzymatic treatment: No enzymatic treatment was performed. |
Column 3: Size Exclusion Chromatography S200 HiLoad 16/60 Superdex run on ÄKTA-Express. |
Column 3 Buffers:
Gel Filtration buffer: 30 mM HEPES pH 7.5, 250 mM NaCl.
|
Column 3 Procedure: Prior to applying the protein, the S200 16/60 column was washed and equilibrated with gel filtration buffer. Eluted protein from the Ni-affinity step was concentrated to 3 ml using a 10 kD MWCO Amicon Ultra concentrator. The concentrated protein was directly applied onto the equilibrated S200 16/60 column, and run at a flow-rate of 1 ml/min. Fractions containing the protein were pooled together, and 10mM DTT was added for overnight storage at 4°C. |
Column 4: Ion Exchange Chromatography Mono Q 1ml run on Äkta Purifier |
Column 4 Buffers:
IEX Buffer A (Binding):50mM HEPES pH7.5.
IEX Buffer B (Elution):50mM HEPES pH7.5, 1M NaCl.
|
Column 4 Procedure: Prior to applying the protein, the mono Q 1ml column was washed with buffer B and then equilibrated in buffer A. Eluted protein from the gel filtration column was diluted to give a final salt concentration below 30mM NaCl. The diluted protein was applied onto the equilibrated mono Q column and the column washed with 2ml of buffer A. Bound proteins were eluted with a gradient up to 40% buffer B. TTC5 eluted between the 80 and 120 mM NaCl range. Clean fractions by SDS PAGE were were pooled. |
Protein concentration: The final buffer was adjusted to 50 mM HEPES, 400 mM NaCl, 10 mM DTT, 50 mM L-Arginine, 50 mM L-Glutamate. The purified protein was concentrated in a 10 kD MWCO Amicon Ultra concentrator to 26 mg/ml (using an estimated extinction coefficient of 48360 M-1 cm-1. |
Mass spectrometry characterization: Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. The intact mass indicated cleavage of the N-terminal methionine and some 50% gluconylation of the His-tag corresponding to experimental masses of 52217.8 and 52395.8 Da (expected MW = 52209.9). |
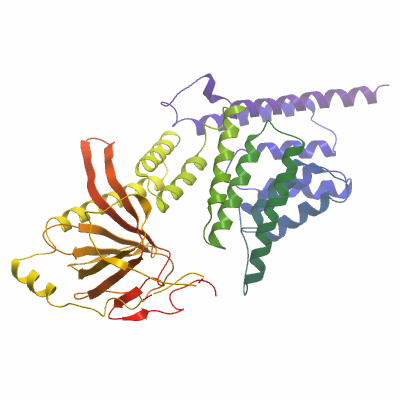
Crystallisation: Protein was crystallized using the sitting drop vapour diffusion technique at 4°C using nanolitre-sized drops. Rod-shaped crystals of native Strap protein appeared in a number of conditions. The best crystals were grown in a 150 nl drop containing equal volumes of protein (26 mg/ml) mixed with well solution (25% tert-butanol, 0.1 M Tris pH 8.5). These crystals were monoclinic and contained two molecules per asymmetric unit (ASU) based on Matthews' coefficient. Monoclinic crystals were also grown for selenomethionine-substituted protein in drops containing 100 nl protein (18 mg/ml) mixed with 50 nl reservoir solution (10% isopropanol, 20% PEG4000, 0.1 M HEPES pH 7.5). |
Data collection:
X-ray source: Swiss Light Source beamline X10SA
Resolution: 2.05Å
Diffraction data were collected on beamline X10SA at the Swiss Light Source (Paul Scherrer Institut, Villigen, Switzerland). Data were collected from crystals at 100K and diffraction images were recorded on a MARCCD 225 detector. Native data were collected to a resolution of 2.05Å. For phasing, a Se-SAD dataset was collected at a wavelength of 0.9789Å. All data were processed with MOSFLM and SCALA from the CCP4 suite of programs.
|