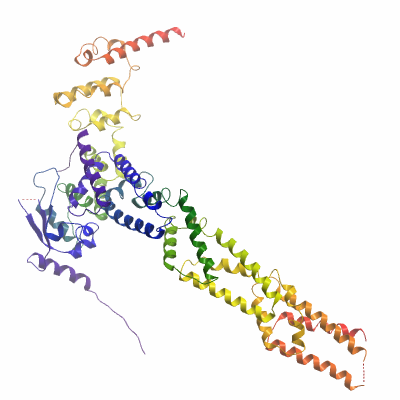
XX06CUL3A (4AP2) Materials & Methods |
KLHL11: Kelch-like protein 11, BTB/BACK domains Material & Methods |
Entry Clone Source: IMAGE |
SGC Construct ID: KLHL11A-c003 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ]. |
DNA sequence:
CATATGCACCATCATCATCATCATTCTTC
TGGTGTAGATCTGGGTACCGAGAACCTGT
ACTTCCAATCCATGGAAGCCGAGGATTTC
GAGTGCAGCTCTCACTGCTCAGAGCTGTC
CTGGCGGCAGAACGAGCAGCGGCGCCAGG
GCCTCTTCTGCGACATTACCCTGTGCTTC
GGCGGGGCTGGAGGCCGCGAGTTCCGGGC
CCACCGCTCGGTACTGGCTGCCGCCACCG
AGTACTTCACGCCCCTGCTCTCGGGCCAG
TTTTCCGAGTCCCGCTCGGGACGGGTGGA
GATGCGCAAGTGGAGCTCCGAGCCGGGGC
CCGAACCCGACACAGTGGAAGCCGTAATC
GAGTACATGTACACCGGGCGCATCCGCGT
CAGCACGGGCAGCGTGCACGAGGTGCTGG
AGTTGGCCGACAGGTTCCTACTCATTCGT
TTAAAAGAATTTTGTGGAGAATTTCTCAA
GAAAAAACTTCATCTCTCAAATTGTGTGG
CAATTCATAGCTTAGCACACATGTACACC
CTGAGCCAACTTGCTCTGAAGGCTGCTGA
TATGATACGGAGAAATTTCCACAAAGTGA
TTCAGGATGAAGAATTTTATACGTTACCT
TTCCATCTCATTAGAGACTGGCTTTCAGA
TTTGGAAATTACAGTTGATTCTGAAGAGG
TTCTCTTTGAAACCGTTTTGAAATGGGTT
CAGAGAAATGCTGAAGAGAGAGAGAGATA
CTTTGAAGAACTTTTTAAATTGCTCAGGT
TGTCCCAGATGAAACCTACCTACCTTACT
CGACATGTCAAACCAGAGAGGCTGGTAGC
CAATAATGAAGTTTGTGTCAAGTTGGTCG
CTGACGCAGTGGAGAGACATGCTCTGAGA
GCTGAGAATATACAATCTGGCACATGACA
GTAAAGGTGGATACGGATCCGAA
|
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfq^sMEA
EDFECSSHCSELSWRQNEQRRQGLFC
DITLCFGGAGGREFRAHRSVLAAATE
YFTPLLSGQFSESRSGRVEMRKWSSE
PGPEPDTVEAVIEYMYTGRIRVSTGS
VHEVLELADRFLLIRLKEFCGEFLKK
KLHLSNCVAIHSLAHMYTLSQLALKA
ADMIRRNFHKVIQDEEFYTLPFHLIR
DWLSDLEITVDSEEVLFETVLKWVQR
NAEERERYFEELFKLLRLSQMKPTYL
TRHVKPERLVANNEVCVKLVADAVER
HALRAENIQSGT
^ TEV protease recognition site
|
Tags and additions: Cleavable N-terminal His6 tag |
Host: BL21(DE3)-R3-pRARE2. |
Growth Medium & Induction Protocol: A glycerol stock was used to inoculate a 10ml starter culture containing LB media with 50µg/ml kanamycin and 34 µg/ml chloramphenicol. The starter culture was grown overnight at 37°C with shaking at 200 rpm. The following morning, four 2L baffled flasks containing 1 L Terrific Broth (TB) media/kanamycin were each inoculated with 2 ml of the starter culture. Cultures were incubated at 37°C with shaking at 180 rpm until an OD600nm = 1.5 was reached. The flasks were then cooled down to 18°C. Protein expression was induced by addition of IPTG to a final concentration of 0.5mM and expression carried out overnight. Cells were harvested by centrifugation at 6000 rpm at 4°C for 15 min. Cell pellets from each flask were resuspended in 30ml binding buffer (50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole), transferred to 50 ml tubes, and stored at -20°C.
|
Extraction buffer, extraction method: The frozen cells were thawed and 0.5mM TCEP, 1mM PMSF added to the cell suspension. The cells were lysed using an Emulsiflex C5 cell homogeniser. The cell lysate was clarified by centrifugation at 20000 rpm at 4°C for 1 h. The supernatant was recovered for purification and filtered through a syringe filter with a 1.2µm pore size.
|
Column 1: Ni-Affinity Chromatography 2.5ml Ni-sepharose slurry applied to a 1.5 x 10 cm column. |
Column 1 Buffers:
Binding buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole
Wash buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 30mM Imidazole
Elution buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 50 to 250mM Imidazole
|
Column 1 Procedure: 2.5ml of 50 % Ni-sepharose slurry (Amersham) was added to the filtered lysate, which was incubated with the Ni-sepharose for 1 hour at 4°C with slow rotation to maximize binding. The lysate was then applied to a 1.5 x 10 cm column by gravity flow and allowed to drip through. The remaining resin was then washed with 2x50ml binding buffer and 1x30ml wash buffer to remove nonspecifically binding proteins. The bound target protein was eluted by applying a step gradient of imidazole (5 ml fractions of elution buffer supplemented with 50mM, 100mM, 150mM and 250mM imidazole). The protein content of collected fractions was visualized using SDS-PAGE and fractions containing KLHL11 were pooled.
|
CUL3: Cullin 3 N-terminal domain Material & Methods |
Entry Clone Source: Synthetic |
SGC Construct ID: CUL3A-c012 |
Vector: pNIC-CTHF. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ]. |
DNA sequence:
CTTAAGAAGGAGATATACTATGAGCAACC
TGTCCAAGGGTACCGGCAGTCGTAAGGAT
ACCAAAATGCGTATCCGCGCCTTCCCGAT
GACTATGGACGAAAAATACGTCAATTCGA
TTTGGGATCTGCTGAAGAACGCTATCCAG
GAGATTCAGCGTAAAAACAATTCTGGTCT
GTCATTTGAAGAGCTGTACCGCAACGCCT
ATACTATGGTGCTGCACAAGCATGGTGAA
AAACTGTATACCGGCCTGCGTGAAGTGGT
CACCGAGCACCTGATCAATAAGGTGCGCG
AGGATGTCCTGAACAGCCTGAATAATAAC
TTCCTGCAGACCCTGAATCAGGCCTGGAA
CGACCATCAGACCGCTATGGTGATGATCC
GTGACATTCTGATGTACATGGATCGCGTT
TATGTGCAGCAGAATAACGTCGAAAATGT
TTACAACCTGGGTCTGATCATTTTTCGTG
ACCAGGTTGTGCGCTATGGCTGCATCCGT
GACCACCTGCGCCAGACCCTGCTGGATAT
GATTGCCCGTGAACGCAAGGGCGAGGTCG
TTGATCGTGGCGCCATCCGCAACGCTTGT
CAGATGCTGATGATTCTGGGTCTGGAAGG
CCGTTCTGTCTACGAAGAGGACTTCGAGG
CGCCCTTTCTGGAAATGAGTGCAGAGTTC
TTTCAGATGGAATCGCAGAAATTCCTGGC
GGAAAACAGCGCATCAGTTTATATCAAGA
AAGTGGAAGCTCGTATCAACGAAGAGATT
GAGCGCGTTATGCATTGCCTGGATAAGTC
AACCGAAGAGCCAATTGTGAAAGTGGTCG
AACGCGAGCTGATCTCTAAGCACATGAAA
ACCATTGTCGAAATGGAAAACAGCGGCCT
GGTTCATATGCTGAAGAACGGTAAAACCG
AAGACCTGGGCTGTATGTACAAGCTGTTC
AGCCGTGTGCCGAATGGTCTGAAAACCAT
GTGCGAATGTATGAGCAGCTATCTGCGTG
AGCAGGGCAAGGCGCTGGTGTCCGAAGAG
GGTGAAGGCAAAAACCCTGTGGACTACCG
CCAGGGTCTGGACGATCTGAAGAGCCGTT
TCGACCGCTTTCTGCTGGAGTCCTTCAAT
AACGATCGTCTGTTTAAACAGACCATTGC
AGGCGACTTCGAATACTTTCTGAATCTGA
ACAGTCGCTCGCCCGAGTATCTGGCAGAG
AACCTCTACTTCCAATCGCACCATCATCA
CCACCATGATTACAAGGATGACGACGATA
AGTGAGGATCC
|
Final protein sequence (Tag sequence in lowercase):
MSNLSKGTGSRKDTKMRIRAFPMTMD
EKYVNSIWDLLKNAIQEIQRKNNSGL
SFEELYRNAYTMVLHKHGEKLYTGLR
EVVTEHLINKVREDVLNSLNNNFLQT
LNQAWNDHQTAMVMIRDILMYMDRVY
VQQNNVENVYNLGLIIFRDQVVRYGC
IRDHLRQTLLDMIARERKGEVVDRGA
IRNACQMLMILGLEGRSVYEEDFEAP
FLEMSAEFFQMESQKFLAENSASVYI
KKVEARINEEIERVMHCLDKSTEEPI
VKVVERELISKHMKTIVEMENSGLVH
MLKNGKTEDLGCMYKLFSRVPNGLKT
MCECMSSYLREQGKALVSEEGEGKNP
VDYRQGLDDLKSRFDRFLLESFNNDR
LFKQTIAGDFEYFLNLNSRSPEYLae
nlyfq^shhhhhhdykddddk
^ TEV protease recognition site
|
Tags and additions: Cleavable C-terminal His6 and FLAG tag |
Host: BL21(DE3)-R3-pRARE2. |
Growth Medium & Induction Protocol: A glycerol stock was used to inoculate a 20ml starter culture containing LB media with 50µg/ml Kanamycin and 34 µg/ml chloramphenicol. The starter culture was grown overnight at 37°C with shaking at 200 rpm. The following morning, six 2L baffled flasks containing 1 L Terrific Broth (TB) media/kanamycin were each inoculated with 2 ml of the starter culture. Cultures were incubated at 37°C with shaking at 180 rpm until an OD600nm = 1.5 was reached. The flasks were then cooled down to 18°C. Protein expression was induced by addition of IPTG to a final concentration of 0.5mM and expression carried out overnight. Cells were harvested by centrifugation at 6000 rpm at 4°C for 15 min. Cell pellets from each flask were resuspended in 30ml binding buffer (50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole), transferred to 50 ml tubes, and stored at -20°C.
|
Extraction buffer, extraction method: The frozen cells were thawed and 0.5mM TCEP, 1mM PMSF added to the cell suspension. The cells were lysed using an Emulsiflex C5 homogeniser. The cell lysate was clarified by centrifugation at 20000 rpm at 4°C for 1 h. The supernatant was recovered for purification and filtered through a syringe filter with 1.2µm pore size.
|
Column 1: Ni-Affinity Chromatography 2.5ml Ni-sepharose slurry applied to a 1.5 x 10 cm column. |
Column 1 Buffers:
Binding buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole
Wash buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 30mM Imidazole
Elution buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 50 to 250mM Imidazole
|
Column 1 Procedure: 2.5ml of 50 % Ni-sepharose slurry (Amersham) was added to the filtered lysate, which was incubated with the Ni-sepharose for 1 hour at 4°C with slow rotation to maximize binding. The lysate was then applied to a 1.5 x 10 cm column by gravity flow and allowed to drip through. The remaining resin was then washed with 2x50ml binding buffer and 1x30ml wash buffer to remove nonspecifically binding proteins. The bound target protein was eluted by applying a step gradient of imidazole (5 ml fractions of elution buffer supplemented with 50mM, 100mM, 150mM and 250mM imidazole). The protein content of collected fractions was visualized using SDS-PAGE and fractions containing CUL3 were pooled.
|
Complex Material & Methods |
Column 1: Size Exclusion Chromatography S200 HiLoad 16/60 Superdex run on ÄKTAxpress. |
Column 1 Buffers:
Gel Filtration buffer: 10mM HEPES pH7.5, 150mM NaCl, 1mM TCEP
|
Column 1 Procedure: Prior to applying the protein, the S200 16/60 column was washed and equilibrated with gel filtration buffer. The two partially purified proteins were combined in an equimolar ratio and concentrated to a volume of 2ml using an Amicon Ultra-15 filter with a 10kDa cut-off. The concentrated protein complex was applied to the equilibrated S200 16/60 column, and run at a flow-rate of 1 ml/min. Fractions were visualized using SDS-PAGE and those containing the complex were pooled.
|
Column 2: Ion Exchange Chromatography HiTrap Q HP 5ml run on Äkta Express |
Column 2 Buffers:
IEX buffer A (binding): 50mM HEPES pH7.5, 50mM NaCl, 0.5mM TCEP
IEX buffer B (elution): 50mM HEPES pH7.5, 500mM NaCl, 0.5mM TCEP
IEX sample buffer: 50mM HEPES pH7.5, 0.5mM TCEP
|
Column 2 Procedure: Prior to applying the protein, the HiTrap Q HP 5ml column was washed with buffer B and equilibrated in buffer A. Eluted protein from the gel filtration column was diluted in sample buffer 10x to give a final buffer concentration of 50mM NaCl. The diluted protein was applied onto the equilibrated HiTrap Q column and the column washed with 4 column volumes of buffer A. Bound proteins were eluted with a gradient up to 50% buffer B over 100ml at 4 ml/minute and 1ml fractions were collected by peak detection. Fractions were visualized using SDS-PAGE and those containing the KLHL11/CUL3 complex were pooled. After concentration, some of the protein was immediately used for crystallization experiments and the rest was divided into 100µl aliquots and stored at -80°C.
|
Mass spec characterization: The purified native complex was homogeneous. KLHL11 had an experimental mass of 34473.3, consistent with the calculated mass of the protein (calculated MW of this species = 34474.3). CUL3 had an experimental mass of 47992.97, consistent with loss of the N-terminal initiator methionine (calculated MW of this species = 48125.1). Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. |
Concentration and complex formulation: The purified complex was buffer exchanged into gel filtration buffer and concentrated to a final concentration of 17mg/ml (measured by OD280 based on extinction coefficient 70250) in an Amicon Ultra-15 filter with a 10 kDa cut-off. |
Crystallization: Initial crystal screens were performed with the full complex at 17 mg/ml. For diffraction-quality crystals, the protein was diluted to 8.5 mg/ml and buffered in 10mM HEPES, pH 7.5, 150mM NaCl, 0.5 mM TCEP. Crystals were grown at 20°C in 300 nl sitting drops mixing 150 nl protein solution with 150 nl of a reservoir solution containing 0.1 M BisTrisPropane pH 7, 25% PEG3350, 0.15M sodium iodide and 8% EthGly. On mounting crystals were cryo-protected with an additional 25% ethylene glycol. |
Data Collection:
Resolution: 2.8Å X-ray source: Diamond IO3
Crystals of the KLHL11/CUL3 complex diffracted to a resolution of 2.8 Å (scaled resolution). A full dataset was collected at 100 K on Diamond Light Source beamline I03. Crystals belonged to the space group I121 with unit-cell parameters a= 147.5 Å b= 40.2 Å c= 234.7 Å, α=90° β= 107.3° γ= 90°. A single complex was present in the asymmetric unit. Data were indexed and integrated using XDS and scaled using SCALA.
Initially, crystals were grown of a complex containing a smaller CUL3 construct with a truncated N-terminus (see SGC deposition target XX01CUL3A). Data were collected to 3.1 Å and experimental phases calculated. Crystals of this KLHL11/CUL3 complex were soaked for 30mins in the crystallization buffer supplemented with 2mM Thiomersal. Data were collected at Diamond Light Source and experimental phases calculated by SIRAS using SHARP. The phased and density-improved map was used to build an initial model using BUCCANEER. This model was further built and refined using COOT and REFMAC5, including the calculated Hendrickson Lattman coefficients as a restraint during refinement. This model (PDB 4APF) was then used as a molecular replacement solution for the larger KLHL11 / CUL3 complex described here (PDB 4AP2), which was completed in COOT and refined with BUSTER.
|