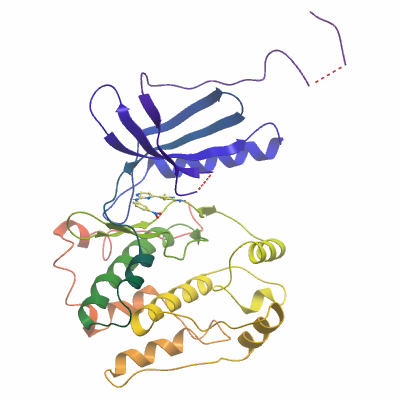
NEK1A (4APC) Materials & Methods |
Entry Clone Source: SGC Oxford |
Entry Clone Accession: gi|19718731 |
Vector: pNIC-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
DNA sequence:
ATGCACCATCATCATCATCATTCTTC
TGGTGTAGATCTGGGTACCGAGAACC
TGTACTTCCAATCCATGGAGAAGTAT
GTTAGACTACAGAAGATTGGAGAAGG
TTCATTTGGAAAAGCCATTCTTGTTA
AATCTACAGAAGATGGCAGACAGTAT
GTTATCAAGGAAATTAACATCTCAAG
AATGTCCAGTAAAGAAAGAGAAGAAT
CAAGGAGAGAAGTTGCAGTATTGGCA
AACATGAAGCATCCAAATATTGTCCA
GTATAGAGAATCATTTGAAGAAAATG
GCTCTCTCTACATAGTAATGGATTAC
TGTGAGGGAGGGGATCTGTTTAAGCG
AATAAATGCTCAGAAAGGCGTTTTGT
TTCAAGAGGATCAGATTTTGGACTGG
TTTGTACAGATATGTTTGGCCCTGAA
ACATGTACATGATAGAAAAATTCTTC
ATCGAGACATTAAATCTCAGAACATA
TTTTTAACTAAAGATGGAACAGTACA
ACTTGGAGATTTTGGAATTGCTAGAG
TTCTTAATAGTACTGTAGAGCTGGCT
CGAGCTTGCATAGGGACCCCATACTA
CTTGTCACCTGAAATCTGTGAAAACA
AACCTTACAATAATAAAAGTGACATT
TGGGCTCTGGGGTGTGTCCTTTATGA
GCTGTGTACACTTAAACATGCTTTTG
AAGCTGGCAGTATGAAAAACCTGGTA
CTGAAGATAATATCTGGATCTTTTCC
ACCTGTGTCTTTGCATTATTCCTATG
ATCTCCGCAGTTTGGTGTCTCAGTTA
TTTAAAAGAAATCCTAGGGATAGACC
ATCAGTCAACTCCATATTGGAGAAAG
GTTTTATAGCCAAACGCATTGAAAAG
TTTCTCTCTCCTCAGCTTATTGCAGA
AGAATTTTGTCTAAAAACATTTTCGA
AGTTTGGATCACAGCCTATACCAGCT
AAAAGACCAGCTTCAGGACAAAACTC
GATTTCTGTTATGCCTGCTCAGAAAA
TTACAAAGCCTGCCGCTAAATATGGA
ATACCTTTAGCATATAAGAAATATGG
AGATAAAAAATGA
|
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfq^sMEK
YVRLQKIGEGSFGKAILVKSTEDGRQ
YVIKEINISRMSSKEREESRREVAVL
ANMKHPNIVQYRESFEENGSLYIVMD
YCEGGDLFKRINAQKGVLFQEDQILD
WFVQICLALKHVHDRKILHRDIKSQN
IFLTKDGTVQLGDFGIARVLNSTVEL
ARACIGTPYYLSPEICENKPYNNKSD
IWALGCVLYELCTLKHAFEAGSMKNL
VLKIISGSFPPVSLHYSYDLRSLVSQ
LFKRNPRDRPSVNSILEKGFIAKRIE
KFLSPQLIAEEFCLKTFSKFGSQPIP
AKRPASGQNSISVMPAQKITKPAAKY
GIPLAYKKYGDKK
^ TEV protease cleavage site
T162A mutation |
Tags and additions: Cleavable N-terminal His6 tag |
Host: BL-21(DE3)-R3-lambda-ppase (A homemade phage resistant version of BL21(DE3) that carries a plasmid for co-expression of lambda phosphatase). |
Growth Medium & Induction Protocol: The construct DNA was transformed into homemade chemically competent cells of the expression strain by a standard heat shock procedure. Colonies were used to inoculate 50 ml of LB media containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol in a 250 ml baffled shaker flask, which was placed in a 37°C shaker overnight. The next day 3x 10 ml of this starter culture was used to inoculate 3x 1L of LB media containing 35 µg/ml kanamycin in 2L baffled shaker flasks. When the OD600 was approximately 0.45, the temperature was reduced to 20°C and when the OD600 was approximately 0.6 the cells were induced by the addition of 0.5 mM IPTG. The expression was continued overnight. Cells were spun at 4000rpm for 10 mins and the pellets resuspended in Lysis Buffer and then frozen at -20°C.
|
Extraction buffer, extraction method: The resuspended cell pellet was thawed and lysed by sonication. PEI (polyethyleneimine) was added to a final concentration of 0.15 %. The cell debris and precipitated DNA were spun down.
Lysis buffer: 50 mM Hepes pH 7.4, 500 mM NaCl, 20 mM Imidazole, 0.5 mM TCEP, 0.2 mM PMSF
|
Column 1: 5 ml of Ni-Sepharose in a 2 cm diameter gravity flow column. |
Column 1 Buffers:
Binding buffer: 50 mM Hepes pH 7.4, 500 mM NaCl, 20 mM Imidazole, 0.5 mM TCEP.
Wash buffer 1: 50 mM Hepes pH 7.4, 1 M NaCl, 40 mM Imidazole, 0.5 mM TCEP.
Wash buffer 2: 50 mM Hepes pH 7.4, 500 mM NaCl, 60 mM Imidazole, 0.5 mM TCEP.
Elution buffer: 50 mM Hepes pH 7.4, 500 mM NaCl, 250 mM Imidazole, 0.5 mM TCEP.
|
Column 1 Procedure: The clarified supernatant was passed through the column. The column was washed with 50 ml of Binding Buffer and 50 ml each of Wash Buffer 1 and 2. 25 ml of Elute Buffer was passed through to elute the protein.
|
Column 2: S200 16/60 Gel Filtration (GE Healthcare) |
Column 2 Buffers:
Gel Filtration buffer: 50 mM Hepes pH 7.4, 300 mM NaCl, 0.5 mM TCEP
|
Column 2 Procedure: The eluted protein was concentrated to 5 ml volume and injected onto the column. |
Mass spec characterization:
Expected: 39850.1
Observed: 39851.4 |
Concentration: The NEK1 was concentrated to 19.8 mg/ml (measured by 280 nm absorbance). |
Crystallization: Crystals grew from a 2:1 ratio of protein and precipitant solution (0.2M ammonium chloride, 20%(w/v) PEG 3350), using the vapour diffusion method. |
Data Collection:
Crystals were cryo-protected by equilibration into precipitant solution containing 25% ethylene glycol, and then flash frozen in liquid nitrogen. Data was collected at Diamond, beamline I24.
|