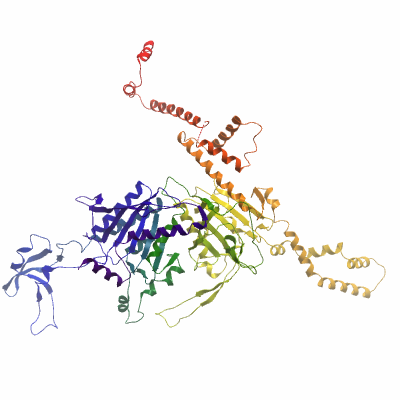
ACACAA (4ASI) Materials & Methods |
Entry clone source: Synthetic |
SGC Construct ID: ACACAA-c106 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ]. |
DNA sequence:
CATATGCACCATCATCATCATCATTC
TTCTGGTGTAGATCTGGGTACCGAGA
ACCTGTACTTCCAATCCATGTTGATC
AACACACCCTATGTCACCAAAGACCT
GTTGCAGAGTAAGCGTTTCCAAGCCC
AGTCGCTGGGTACTACATACATCTAT
GATATCCCCGAAATGTTTCGCCAGAG
CCTGATTAAGTTGTGGGAATCCATGT
CTACCCAAGCATTCCTGCCTTCACCG
CCCTTGCCAAGTGACATGCTGACTTA
CACCGAGCTGGTATTGGATGACCAGG
GCCAACTGGTGCACATGAATCGCCTC
CCCGGAGGCAACGAAATCGGTATGGT
TGCGTGGAAGATGACTTTTAAGTCGC
CGGAGTATCCCGAAGGCCGCGATATC
ATTGTCATCGGTAATGACATTACGTA
TCGCATCGGATCGTTTGGTCCACAAG
AGGATCTGTTGTTCCTGCGCGCATCC
GAACTGGCCCGTGCTGAGGGCATTCC
TCGCATCTACGTATCTGCGAACTCAG
GTGCCCGTATTGGCTTGGCAGAAGAG
ATCCGCCACATGTTTCACGTGGCTTG
GGTTGACCCGGAAGATCCATATAAAG
GTTACCGTTATCTGTACTTGACTCCC
CAAGACTATAAGCGCGTCAGTGCCCT
GAACTCGGTGCATTGTGAGCACGTAG
AAGATGAAGGCGAGAGCCGTTACAAA
ATCACAGACATTATCGGTAAGGAAGA
GGGCATTGGTCCTGAAAATCTGCGCG
GCTCCGGTATGATCGCGGGCGAGTCG
TCATTGGCATATAATGAAATTATCAC
CATTAGTCTGGTGACTTGCCGTGCTA
TCGGTATCGGCGCCTACTTGGTTCGC
CTGGGCCAGCGTACCATTCAGGTCGA
AAATTCGCATTTGATCCTGACAGGCG
CGGGTGCTCTGAACAAAGTATTGGGC
CGCGAAGTGTACACTAGCAACAATCA
ACTGGGCGGCATTCAGATCATGCACA
ACAATGGTGTTACCCATTGTACAGTC
TGCGATGACTTCGAGGGCGTGTTTAC
CGTATTGCACTGGCTGTCCTATATGC
CGAAATCTGTTCATTCAAGTGTCCCA
CTGTTGAACAGCAAGGACCCCATTGA
CCGTATCATTGAGTTCGTGCCTACTA
AAACCCCGTACGATCCACGCTGGATG
CTGGCCGGTCGTCCCCACCCTACACA
AAAGGGTCAGTGGTTGTCCGGCTTCT
TCGACTATGGTTCGTTCTCTGAGATC
ATGCAGCCGTGGGCACAAACTGTGGT
AGTTGGCCGCGCGCGTCTGGGCGGCA
TCCCAGTCGGTGTGGTAGCTGTTGAA
ACCCGCACAGTCGAGTTGTCAATTCC
CGCCGATCCTGCAAACCTGGACAGTG
AAGCCAAAATCATTCAGCAAGCGGGC
CAGGTGTGGTTTCCGGATAGCGCTTT
CAAAACGTATCAGGCCATCAAGGACT
TTAATCGTGAAGGTCTGCCATTGATG
GTGTTCGCAAACTGGCGCGGCTTTTC
CGGCGGCATGAAAGATATGTATGACC
AGGTACTGAAGTTCGGTGCGTACATT
GTTGATGGCTTGCGTGAGTGTTGCCA
GCCCGTCCTGGTGTATATCCCTCCGC
AAGCTGAATTGCGCGGCGGCTCGTGG
GTAGTTATTGACTCTTCAATCAATCC
ACGTCACATGGAGATGTACGCCGATC
GCGAAAGCCGTGGTAGTGTCCTGGAG
CCAGAAGGCACCGTGGAGATCAAATT
CCGCCGTAAAGACCTGGTAAAGACCA
TGCGCCGTGTTGATCCTGTCTACATT
CACTTGGCAGAACGCCTGGGTACACC
GGAGTTGTCGACGGCAGAACGTAAAG
AGCTGGAAAACAAGCTGAAAGAGCGC
GAAGAGTTCTTGATCCCAATCTATCA
TCAAGTGGCTGTGCAATTCGCCGACC
TGCACGATACCCCCGGCCGTATGCAG
GAGAAAGGTGTAATCTCGGACATTTT
GGATTGGAAAACATCTCGCACTTTCT
TCTACTGGCGTCTGCGCCGTTTGCTG
CTGGAGGACTTGGTTAAAAAAAAGAT
CCATAATGCAAACCCTGAGTTGACCG
ATGGCCAAATTCAGGCGATGTTGCGC
CGTTGGTTTGTCGAAGTGGAGGGTAC
AGTAAAGGCTTATGTTTGGGACAACA
ATAAGGATCTGGCCGAATGGCTGGAG
AAACAGTTGACTGAAGAGGACGGCGT
CCACTCAGTGATCGAAGAGAACATTA
AGTGTATCAGCCGCGATTACGTACTG
AAGCAAATCCGTTCCTTGGTTCAGGC
AAATCCGGAAGTCGCGATGGACAGTA
TTATCCACATGACCCAACACATTTCT
CCAACACAGCGCGCTGAGGTGATCCG
TATTCTGTGACAGTAAAGGTGGATAC
GGATCCGAA
|
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfq^smLI
NTPYVTKDLLQSKRFQAQSLGTTYIY
DIPEMFRQSLIKLWESMSTQAFLPSP
PLPSDMLTYTELVLDDQGQLVHMNRL
PGGNEIGMVAWKMTFKSPEYPEGRDI
IVIGNDITYRIGSFGPQEDLLFLRAS
ELARAEGIPRIYVSANSGARIGLAEE
IRHMFHVAWVDPEDPYKGYRYLYLTP
QDYKRVSALNSVHCEHVEDEGESRYK
ITDIIGKEEGIGPENLRGSGMIAGES
SLAYNEIITISLVTCRAIGIGAYLVR
LGQRTIQVENSHLILTGAGALNKVLG
REVYTSNNQLGGIQIMHNNGVTHCTV
CDDFEGVFTVLHWLSYMPKSVHSSVP
LLNSKDPIDRIIEFVPTKTPYDPRWM
LAGRPHPTQKGQWLSGFFDYGSFSEI
MQPWAQTVVVGRARLGGIPVGVVAVE
TRTVELSIPADPANLDSEAKIIQQAG
QVWFPDSAFKTYQAIKDFNREGLPLM
VFANWRGFSGGMKDMYDQVLKFGAYI
VDGLRECCQPVLVYIPPQAELRGGSW
VVIDSSINPRHMEMYADRESRGSVLE
PEGTVEIKFRRKDLVKTMRRVDPVYI
HLAERLGTPELSTAERKELENKLKER
EEFLIPIYHQVAVQFADLHDTPGRMQ
EKGVISDILDWKTSRTFFYWRLRRLL
LEDLVKKKIHNANPELTDGQIQAMLR
RWFVEVEGTVKAYVWDNNKDLAEWLE
KQLTEEDGVHSVIEENIKCISRDYVL
KQIRSLVQANPEVAMDSIIHMTQHIS
PTQRAEVIRIL
^ TEV protease recognition site
|
Tags and additions: Cleavable N-terminal His6 tag |
Host: BL21(DE3)-R3-pRARE2. Phage-resistant strain. |
Growth Medium & Induction Protocol: One colony from the transformation was used to inoculate 1 ml of TB media containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol, which was placed in a 37°C shaker overnight. The next day glycerol stocks were prepared from this overnight culture.
A glycerol stock was used to inoculate 50 ml of TB media containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol, which was placed in a 37°C shaker overnight. The next day this starter culture was used to inoculate 12L of TB media (7.5 ml starter culture used per 1L) containing 50 µg/ml kanamycin. When the OD600 reached approximately 1.0 the temperature was reduced to 18°C and after a further 30 minutes the cells were induced by the addition of 0.1 mM IPTG. The expression was continued overnight.
Cells were harvested by centrifugation at 6000 x g after which the supernatant was poured out and the cell pellet either placed in a -20°C freezer or used directly for purification.
|
Cell Lysis: 12,000 psi for 5 passes. The cell debris was pelleted at 35,000 x g and the supernatant used for further purification.
Lysis buffer: 50 mM HEPES pH 7.4, 500 mM NaCl, 5% Glycerol, 10 mM Imidazole pH 7.4, 0.5 mM TCEP, 1 tablet per 50 ml protease inhibitor cocktail EDTA-free (Roche)
|
Column 1: Ni-NTA (2.5 ml volume in a gravity-flow column). |
Column 1 Buffers:
Binding buffer: 50 mM HEPES pH 7.4, 500 mM NaCl, 5% Glycerol, 10 mM Imidazole pH 7.4, 0.5 mM TCEP
Wash buffer: 50 mM HEPES pH 7.4, 500 mM NaCl, 5% Glycerol, 40 mM Imidazole pH 7.4, 0.5 mM TCEP
Elution buffer: 50 mM HEPES pH 7.4, 500 mM NaCl, 5% Glycerol, 250 mM Imidazole pH 7.4, 0.5 mM TCEP
|
Column 1 Procedure: The clarified cell extract was incubated with 2.5 ml of Ni-NTA pre-equilibrated with lysis buffer for 1 hour at 4°C with rotation after which it was passed through a glass column. The column was then washed with Binding Buffer (60 ml) and Wash Buffer (50 ml). The protein was eluted with 25 ml of Elution Buffer in 5 x 5 ml fractions.
|
Column 2: Superdex 200 16/60 Gel Filtration |
Column 2 Buffers:
Gel Filtration buffer: 10 mM Hepes pH 7.4, 500 mM NaCl, 0.5 mM TCEP, 5% Glycerol
|
Column 2 Procedure: The wash buffer fractions and elution buffer fractions from column 1 were pooled separately and concentrated to 5 ml with a 30 kDa mwco spin concentrator and injected onto an s200 16/60 column (pre-equilibrated in GF Buffer) at 1.0 ml/min. 1.0 ml fractions were collected. The protein eluted at between 85 ml and 95 ml volume.
|
Column 3: TEV cleavage/ Ni-NTA rebind |
Column 3 Procedure: Protein from fractions eluted at 80-90 ml from s200 gel filtration were pooled and incubated with 1:20 mol:mol TEV protease overnight at 4°C. Then protein plus TEV was passed through a column containing 0.5 ml Ni-NTA pre-equilibrated with GF Buffer. Column was washed 1ml of GF Buffer. Flow-through was pooled.
|
Column 4: Ni-NTA (2.5 ml volume in a gravity-flow column). |
Column 4 Buffers:
Buffer A: 50 mM HEPES pH 7.5, 50 mM NaCl, 5% glycerol
Buffer B: 50 mM HEPES pH 7.5, 2 M NaCl, 5% glycerol
Buffer C: 50 mM HEPES pH7.5, 5% glycerol
|
Column 4 Procedure: Protein from flow-through, approximately 20 ml, was concentrated to 5 ml using a 30 kDa mwco spin concentrator, diluted to 50 ml using Buffer C and injected into a 5 ml HiTrap Q column. Protein was eluted using a linear gradient of 0-25 % Buffer B over 30 column volumes at 2ml/min. 1.5 ml fractions were collected.
|
Concentration: Protein eluted at 6-9% Buffer B were pooled and concentrated to 28 mg/ml using a 30 kDa mwco concentrator.
|
Mass spec characterization:
Expected mass: 87398.3 Da, Measured mass: 87406.8 Da
|
Crystallization: Crystals were grown by vapour diffusion in sitting drop at 20°C by setting up 12mg/ml of protein in the presence of 1mM malonyl-CoA. A sitting drop consisting of 75 nl protein and 75 nl well solution was equilibrated against well solution containing 19% (v/v) PEG 3350 and 0.05 M sodium malonate. Crystals were mounted in the presence of 25% (v/v) ethylene glycol and flash-cooled in liquid nitrogen. |
Data Collection: Resolution: 2.8 Å
X-ray source: Diamond Light Source beamline IO2
|