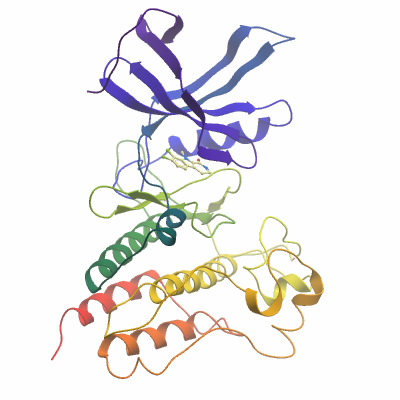
ACVR2A (4ASX) Materials & Methods |
Entry clone source: Origene |
Entry clone accession: GI:65508448 |
SGC Construct ID: ACVR2A-c046 |
Vector: pFB-LIC-Bse. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ]. |
DNA sequence:
TACTTCCAATCCATGCCACTGCAGTT
ATTAGAAGTGAAAGCAAGGGGAAGAT
TTGGTTGTGTCTGGAAAGCCCAGTTG
CTTAACGAATATGTGGCTGTCAAAAT
ATTTCCAATACAGGACAAACAGTCAT
GGCAAAATGAATACGAAGTCTACAGT
TTGCCTGGAATGAAGCATGAGAACAT
ATTACAGTTCATTGGTGCAGAAAAAC
GAGGCACCAGTGTTGATGTGGATCTT
TGGCTGATCACAGCATTTCATGAAAA
GGGTTCACTATCAGACTTTCTTAAGG
CTAATGTGGTCTCTTGGAATGAACTG
TGTCATATTGCAGAAACCATGGCTAG
AGGATTGGCATATTTACATGAGGATA
TACCTGGCCTAAAAGATGGCCACAAA
CCTGCCATATCTCACAGGGACATCAA
AAGTAAAAATGTGCTGTTGAAAAACA
ACCTGACAGCTTGCATTGCTGACTTT
GGGTTGGCCTTAAAATTTGAGGCTGG
CAAGTCTGCAGGCGATACCCATGGAC
AGGTTGGTACCCGGAGGTACATGGCT
CCAGAGGTATTAGAGGGTGCTATAAA
CTTCCAAAGGGATGCATTTTTGAGGA
TAGATATGTATGCCATGGGATTAGTC
CTATGGGAACTGGCTTCTCGCTGTAC
TGCTGCAGATGGACCTGTAGATGAAT
ACATGTTGCCATTTGAGGAGGAAATT
GGCCAGCATCCATCTCTTGAAGACAT
GCAGGAAGTTGTTGTGCATAAAAAAA
AGAGGCCTGTTTTAAGAGATTATTGG
CAGAAACATGCTGGAATGGCAATGCT
CTGTGAAACCATTGAAGAATGTTGGG
ATCACGACGCAGAAGCCAGGTTATCA
GCTGGATGTGTAGGTGAAAGAATTAC
CCAGATGCAGAGACTAACAAATATTT
GACAGTAAAGGTGGATA
|
Expressed protein sequence (His6 affinity Tag sequence in lowercase):
mghhhhhhssgvdlgtenlyfq^SMP
LQLLEVKARGRFGCVWKAQLLNEYVA
VKIFPIQDKQSWQNEYEVYSLPGMKH
ENILQFIGAEKRGTSVDVDLWLITAF
HEKGSLSDFLKANVVSWNELCHIAET
MARGLAYLHEDIPGLKDGHKPAISHR
DIKSKNVLLKNNLTACIADFGLALKF
EAGKSAGDTHGQVGTRRYMAPEVLEG
AINFQRDAFLRIDMYAMGLVLWELAS
RCTAADGPVDEYMLPFEEEIGQHPSL
EDMQEVVVHKKKRPVLRDYWQKHAGM
AMLCETIEECWDHDAEARLSAGCVGE
RITQMQRLTNI
^ TEV protease recognition site
|
Tags and additions: Tev-cleavable N-terminal His6 tag |
Host: SF9 Spodoptera frugiperda Insect cells. |
Growth medium, induction protocol: 2L of SF9 cells at a density of 2 million/ml were infected with 10 ml of Virus/L.Cells were incubated at 27°C in the shaker incubator and harvested after 48 hours. Cells were harvested by centrifugation at 900g at 4°C for 20 min. Cell pellets from each flask (1l volume) were resuspended in 20 ml binding buffer (50 mM Hepes, pH 7.5; 500 mM NaCl; 5% Glycerol; 5 mM imidazole) in the presence of protease inhibitor SET V (Calbiochem) added to the cell suspension at 1:100 dilution, transferred to 50 ml tubes, and stored at 20°C.
|
Extraction buffer, extraction method: The frozen cells were thawed and 0.15% PEI (pH7.4) was added. The cells were lysed by passage 4-5x through an Emulsiflex C5 homogenizer. The cell lysate was spun down by centrifugation at 21.5K rpm at 4°C for 1 h. The supernatant was recovered for purification
|
Column 1: Ni-Affinity Chromatography. 4 ml of 50 % Ni-sepharose slurry was applied onto a 1.5 x 10 cm column. The column was equilibrated with binding buffer (25ml). |
Column 1 Buffers:
Binding buffer: 50 mM Hepes, pH 7.5; 500 mM NaCl; 5% Glycerol; 5 mM imidazole0.5mM TCEP
Wash buffer: 50 mM Hepes, pH 7.5; 500 mM NaCl; 5% Glycerol; 25 mM imidazole0.1mM TCEP
Elution buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% Glycerol; 50 to 250 mM imidazole 0.5mM TCEP
|
Column 1 Procedure: The supernatant following centrifugation was applied by gravity flow onto the Ni-sepharose column. The bound protein was then washed with 50ml binding buffer and subsequently with 30 ml wash buffer. ACVR2A protein was then eluted by applying a step gradient of imidazole using 5 ml portions of elution buffer with increasing concentration of imidazole (50 mM, 100 mM, 150 mM, 250 mM). Elution fractions were analyzed by SDS PAGE and the 100, 150 and 250 mM imidazole fractions were kept and pooled. 10 mM DTT was added for overnight storage at 4°C.
|
Enzymatic treatment: The tag was not cleaved |
Column 2: Size Exclusion Chromatography S200 HiLoad 16/60 Superdex run on ÄKTA-Express |
Column 2 Buffers:
Gel Filtration buffer: 300 mM NaCl, 50 mM HEPES pH 7.5, 0.5 mM TCEP, pH 7.5
|
Column 2 Procedure: Prior to applying the protein, the S200 16/60 column was washed and equilibrated with gel filtration buffer. The protein was concentrated to 3 ml using an Amicon Ultra-15 filter with a 30 kDa cut-off. The concentrated protein was directly applied onto the equilibrated S200 16/60 column, and run at a flow-rate of 1 ml/min. Fractions containing the protein were pooled together.
|
Mass spec characterization:
The purified protein was homogeneous and had an experimental mass of 36410.6 Da. The theoretical expected mass from the construct primary sequence is 36497.9. The difference corresponds to loss of the N-terminal methionine and acetylation of the resulting N-terminus. Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. Proteins were desalted prior to mass spectrometry by rapid elution off a C3 column with a gradient of 5-95% methanol in water with 0.1% formic acid.
|
Concentration: Protein was concentrated to 12.4 mg/ml using an Amicon 30 kDa cut-off concentrator.
|
Crystallization: Protein was concentrated to 12.4 mg/ml in gel filtration buffer. The oxo-beta-carboline inhibitor Dihydro-Bauerine C (K01764) was added to the final sample at a concentration of 1mM. Crystals were grown at 4°C in 150 nl sitting drops mixing 100 nl protein solution with 50 nl of a reservoir solution containing 20% PEG 3350; 0.20M Na(malonate). On mounting crystals were cryo-protected with an additional 20% ethylene glycol. |
Data Collection: Resolution: 2.05 Å
X-ray source: Diamond Light Source, station I04, using monochromatic radiation at wavelength 0.9611 Å
|