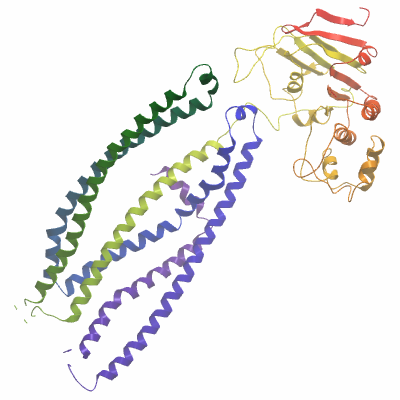
ABCB10A (4AYW) Materials & Methods |
Entry Clone Source: MGC |
Entry Clone Accession: IMAGE: 6143235 |
SGC Construct ID: ABCB10A-c007 |
GI Number: 9961244 |
Vector: pFB-LIC-Bse. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
DNA sequence:
ATGCGAGGCCCCCCTGCCTGGGCAGG
GGACGAGGCCTGGCGGCGCGGGCCGG
CGGCGCCTCCCGGGGACAAGGGGCGG
CTGCGCCCCGCAGCGGCCGGACTCCC
GGAGGCCCGGAAGCTCCTGGGGCTGG
CGTACCCTGAGCGCCGGAGGCTGGCA
GCTGCGGTTGGATTTCTCACGATGTC
CAGTGTTATCTCCATGTCTGCCCCTT
TCTTCCTGGGGAAGATCATTGATGTC
ATCTATACCAACCCCACTGTGGACTA
CAGCGACAACCTGACCCGCCTCTGCC
TAGGGCTCAGTGCCGTGTTTCTGTGT
GGTGCTGCCGCCAATGCCATTCGTGT
CTACCTCATGCAAACTTCAGGTCAGC
GCATTGTGAATAGGCTGAGAACTTCA
TTATTCTCCTCCATTCTGAGGCAGGA
GGTTGCTTTCTTTGACAAGACTCGCA
CAGGAGAATTGATTAACCGCCTCTCA
TCAGACACTGCACTCCTGGGGCGCTC
AGTGACTGAAAACCTCTCAGATGGGC
TCAGGGCCGGGGCCCAGGCTTCTGTA
GGCATCAGTATGATGTTTTTTGTCTC
ACCTAATCTGGCCACCTTTGTTTTGA
GCGTGGTGCCTCCAGTGTCAATCATT
GCTGTAATTTATGGGCGATATCTACG
GAAACTGACCAAAGTCACTCAGGATT
CCCTGGCACAAGCCACTCAGCTAGCT
GAGGAACGTATTGGAAATGTAAGAAC
TGTTCGAGCTTTTGGGAAAGAAATGA
CTGAAATCGAGAAATATGCCAGCAAA
GTGGACCATGTAATGCAGTTAGCAAG
GAAAGAGGCATTCGCCCGGGCTGGTT
TCTTTGGAGCAACTGGGCTCTCCGGA
AACCTGATCGTGCTTTCTGTCCTGTA
CAAAGGAGGGCTGCTGATGGGCAGTG
CCCACATGACCGTGGGTGAACTCTCT
TCCTTCCTAATGTATGCTTTCTGGGT
TGGAATAAGCATTGGAGGTCTGAGCT
CTTTCTACTCGGAGCTGATGAAAGGA
CTGGGTGCAGGGGGGCGCCTCTGGGA
GCTCCTGGAGAGAGAGCCCAAGCTGC
CTTTTAACGAGGGGGTCATCTTAAAT
GAGAAAAGCTTCCAGGGTGCTTTGGA
GTTTAAGAACGTGCATTTTGCCTATC
CAGCTCGCCCAGAGGTGCCCATATTT
CAGGATTTCAGCCTTTCCATTCCGTC
AGGATCTGTCACGGCACTGGTTGGCC
CAAGTGGTTCTGGCAAATCAACAGTG
CTTTCACTCCTGCTGAGGTTGTACGA
CCCTGCTTCTGGAACTATTAGTCTTG
ATGGCCATGACATCCGTCAGCTAAAC
CCAGTGTGGCTGAGATCCAAAATTGG
GACAGTGAGTCAGGAACCCATTTTGT
TTTCTTGCTCTATTGCTGAGAACATT
GCTTATGGTGCTGATGACCCTTCCTC
TGTGACCGCTGAGGAAATCCAGAGAG
TGGCTGAAGTGGCCAATGCAGTGGCC
TTCATCCGGAATTTCCCCCAAGGGTT
CAACACTGTGGTTGGAGAAAAGGGTG
TTCTCCTCTCAGGTGGGCAGAAACAG
CGGATTGCGATTGCCCGTGCTCTGCT
AAAGAATCCCAAAATTCTTCTCCTAG
ATGAAGCAACCAGTGCGCTGGATGCC
GAAAATGAGTACCTTGTTCAAGAAGC
TCTAGATCGACTGATGGATGGAAGAA
CGGTGTTAGTTATTGCCCATCATCTG
TCCACCATTAAGAATGCTAATATGGT
TGCTGTTCTTGACCAAGGAAAAATTA
CTGAATATGGAAAACATGAAGAGCTG
CTTTCAAAACCAAATGGGATATACAG
AAAACTAATGAACAAACAAAGTTTTA
TTTCAGCATGA
|
Expressed sequence (small letters refer to tag sequence):
mghhhhhhssgvdlgtenlyfqs^MR
GPPAWAGDEAWRRGPAAPPGDKGRLR
PAAAGLPEARKLLGLAYPERRRLAAA
VGFLTMSSVISMSAPFFLGKIIDVIY
TNPTVDYSDNLTRLCLGLSAVFLCGA
AANAIRVYLMQTSGQRIVNRLRTSLF
SSILRQEVAFFDKTRTGELINRLSSD
TALLGRSVTENLSDGLRAGAQASVGI
SM MFFVSPNLATFVLSVVPPVSIIA
VIYGRYLRKLTKVTQDSLAQATQLAE
ERIGNVRTVRAFGKEMTEIEKYASKV
DHVMQLARKEAFARAGFFGATGLSGN
LIVLSVLYKGGLLMGSAHMTVGELSS
FLMYAFWVGISIGGLSSFYSELMKGL
GAGGRLWELLEREPKLPFNEGVILNE
KSFQGALEFKNVHFAYPARPEVPIFQ
DFSLSIPSGSVTALVGPSGSGKSTVL
SLLLRLYDPASGTISLDGHDIRQLNP
VWLRSKIGTVSQEPILFSCSIAENIA
YGADDPSSVTAEEIQRVAEVANAVAF
IRNFPQGFNTVVGEKGVLLSGGQKQR
IAIARALLKNPKILLLDEATSALDAE
NEYLVQEALDRLMDGRTVLVIAHHLS
TIKNANMVAVLDQGKITEYGKHEELL
SKPNGIYRKLMNKQSFISA
|
Tags and additions: N-terminal, TEV cleavable hexahistidine tag. (^ cleavage site) |
Host: Spodoptera frugiperda (SF9) insect cells |
Growth Medium & Induction Protocol: Insect cells with a density of 2x106 per litre of cell culture in SF900 medium (Invitrogen) were infected with recombinant baculovirus (5ml P2 virus per litre cell culture) and incubated for 72 hours. Cells were harvested by centrifugation and the pellet was flash frozen in liquid N2.
|
Extraction buffer, extraction method: Frozen pellets were thawed and re-suspended in hypotonic buffer for lysis using a dounce homogeniser and DIAX homogeniser (Heidolph) at 10,000 r.p.m for 2 minutes. Cell membranes were collected by ultracentrifugation at 100,000Xg and the homogenisation repeated. The membranes were further washed with hypertonic buffer twice and the washed membranes were re-suspended in extraction buffer supplemented with 1% DDM, 0.1% CHS and incubated at 4°C with stirring. The extracted protein was separated from insoluble membranes by ultracentrifugation and collected for purification.
|
Hypotonic buffer: 10mM HEPES pH7.5, 0.5 mM EDTA
Hypertonic buffer: 10mM HEPES pH7.5,1M NaCl, 0.5mM EDTA
Extraction/wash buffer: 50 mM HEPES, pH 7.5; 200 mM NaCl; 20 mM imidazole, 0.5mM MgCl2 and 0.5 mM TCEP |
Column 1: Co-affinity. Cobalt Talon (Clontech), 4 ml of 50 % slurry in 1.5 x 10 cm column, washed with extraction/ wash buffer. |
Column 1 Buffers:
Extraction/wash buffer: 50 mM HEPES, pH 7.5; 200 mM NaCl; 20 mM imidazole, 0.5mM MgCl2 and 0.5 mM TCEP ,0.02%DDM, 0.002%CHS
Elution buffer: 50 mM HEPES, pH 7.5; 200 mM NaCl; 300 mM imidazole, 0.5mM MgCl2 and 0.5 mM TCEP, 0.02%DDM, 0.002%CHS
|
Column 1 Procedure: The solubilised membrane protein was batch bound to Cobalt Talon resin equilibrated with extraction buffer at 4°C for one hour and subsequently poured into a gravity flow glass column. The column was then washed with 25CV of wash buffer followed by elution in 1CV fractions until all the protein was eluted.
|
Enzymatic Treatment: 1mg TEV protease was added overnight at 4°C to purified protein with concurrent buffer exchange by dialysis using a 3kDa cut off dialysis membrane into gel filtration buffer. The protease was removed and tag cleaved protein was collected by binding to Cobalt talon and collecting the flow-through. |
Column 2: Size Exclusion Chromatography. Superose6 (GE healthcare) |
Column 2 Buffer:
Dialysis/ Gel Filtration buffer: 20 mM HEPES pH 7.5, 200 mM NaCl, 0.5 mM TCEP, 0.5mM MgCl2, 0.02% DDM+0.001%Cardiolipin
|
Column 2 Procedure: The protein was concentrated and applied to a Superose6 gel filtration column equilibrated with gel filtration buffer using an AKTA Prime system.
|
Mass spec characterization:
The purified protein was homogeneous and had an experimental mass of 67075 Da. This represents an unexplained, average mass of +91Da when compared to the calculated mass (66984 Da) based on the known sequence. Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. Proteins were desalted prior to mass spectrometry by rapid elution off a C3 column with a gradient of 5-95% methanol in water with 0.1% formic acid.
|
Protein Concentration: Protein was concentrated to >8mg/ml using a 100kDa cut-off concentrator and back diluted to 5-8mg/ml with the addition of 2mM Mg-AMP-PNP. |
Crystallization: Crystals were grown in sitting drops at 20°C.
Drops (200nl) comprising protein solution (5mg/ml; ABCB10A DDM/CHS/AMP-PNP) and reservoir solution (0.1-0.2 M NaCl, 5-7% (v/v) jeffamine M600, 30-40% (v/v) PEG400, 0.1 M glycine pH 9.5) in protein:reservoir ratios of 2:1 or 1.5:1 were equilibrated against 20µl of the same reservoir solution.
Crystal plates were transferred to 6°C prior to directly flash cooling crystals in liquid nitrogen. |
Data Collection:
Resolution: 3.3Å
Data were collected at 100°K using a spiral scan collection strategy and a 10x50µm beamsize on I24 microfocus beamline (Diamond Light Source, UK).
Native data were collected to 3.4Å from a single crystal (Λ=0.9686Å). Data for a mercury derivative, prepared by soaking native crystals overnight with 1mM EMTS, were collected to 4Å (µ=0.9779Å). A higher resolution native dataset (nominally 3.15Å) was collected from a crystal soaked for 5mins in mother liquor containing 10mM lutetium chloride.
|
Structure Solution: Initial phases were calculated using SIRAS. Two Hg sites were located with SHELXD and phase refinement was carried out with SHARP / SOLVE. Phase extension / density modification to 3.4Å yielded an interpretable map and allowed an initial backbone trace to be built. However, both data anisotropy and disorder of the NBD hindered model completion using the plate form crystals. The structure was finally solved using the refined, higher resolution rod-form A structure. Molecular replacement was used to position the separate transmembrane and nucleotide-binding domains. Refinement was carried out with autoBUSTER using LSSR restraints to the higher resolution rod-form A structure.
|