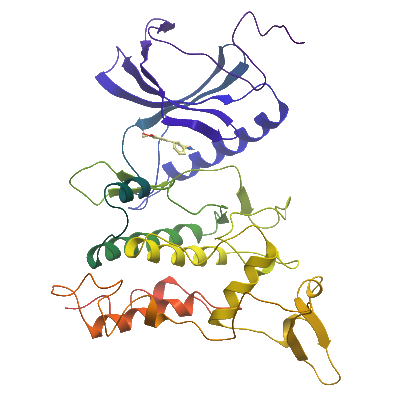DYRK1A belongs to the dual-specificity tyrosine phosphorylation-regulated kinase (DYRK)/Minibrain family of protein kinases. Members of this family share a high degree of conservation in the catalytic domain and in the adjacent N-terminal DH box (DYRK homology), but are very divergent in their N- and C-terminal domains. The DYRK family is distinguished from other kinases by its activation mechanism. DYRK kinase activity depends on tyrosine autophosphorylation at a conserved YxY site in the catalytic domain. This activation domain was shown to be intramolecularly phosphorylated only during translation while mature (phosphorylated) members of the DYRK family have only serine/threonine kinase activity (Friedman, 2007). Activity of DYRK1A activity can be further increased by binding to 14- 3-3 proteins (Kim et al., 2004), mediated by autophosphorylation at a C-terminal serine.
The DYRK1A gene is localized in the Down syndrome critical region of chromosome 21, and is thought to be a strong candidate gene for learning defects associated with Down syndrome. Several transgenic mouse models have been generated supporting this hypothesis, which are found to exhibit neurodevelopmental delays and impairment in learning tasks (Ahn et al., 2006; Altafaj et al., 2001; Smith et al., 1997). A 1.5-fold increase in dosage of DSCR1 and DYRK1A cooperatively destabilize a regulatory circuit, leading to reduced NFATc activity and many of the features of Down's syndrome (Arron et al., 2006). DYRK1A has been found to be over expressed 1.5 times in DS fetal brains.
Here we report the structure of the DYRK1A kinase domain in complex with Leucettine L41, an optimized analogue of the marine sponge natural product Leucettamine B, at 3.15α resolution.
The inhibitor was kindly provided by the laboratory of Laurent Meijer, CNRS, Roscoff, France.