Entry Clone Source: TKC |
Entry Clone Accession: n/a |
SGC Construct ID: STK10A-c013 |
GenBank GI number: gi|5174701 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Tags and additions: Tag sequence:
mhhhhhhssgvdlgtenlyfq*s(m) TEV-cleavable (*) N-terminal his6 tag. |
Amplified construct sequence:
ATGCACCATCATCATCATCATTCTTC
TGGTGTAGATCTGGGTACCGAGAACC
TGTACTTCCAATCCATGAGAAAGTCC
CGCGAATATGAGCACGTCCGCCGCGA
CCTGGACCCCAACGAGGTGTGGGAGA
TCGTGGGCGAGCTGGGCGACGGCGCC
TTCGGCAAGGTTTACAAGGCCAAGAA
TAAGGAGACGGGTGCTTTGGCTGCGG
CCAAAGTCATTGAAACCAAGAGTGAG
GAGGAGCTGGAGGACTACATCGTGGA
GATTGAGATCCTGGCCACCTGCGACC
ACCCCTACATTGTGAAGCTCCTGGGA
GCCTACTATCACGACGGGAAGCTGTG
GATCATGATTGAGTTCTGTCCAGGGG
GAGCCGTGGACGCCATCATGCTGGAG
CTGGACAGAGGCCTCACGGAGCCCCA
GATACAGGTGGTTTGCCGCCAGATGC
TAGAAGCCCTCAACTTCCTGCACAGC
AAGAGGATCATCCACCGAGATCTGAA
AGCTGGCAACGTGCTGATGACCCTCG
AGGGAGACATCAGGCTGGCTGACTTT
GGTGTGTCTGCCAAGAATCTGAAGAC
TCTACAGAAACGAGATTCCTTCATCG
GCACGCCTTACTGGATGGCCCCCGAG
GTGGTCATGTGTGAGACCATGAAAGA
CACGCCCTACGACTACAAAGCCGACA
TCTGGTCCCTGGGCATCACGCTGATT
GAGATGGCCCAGATCGAGCCGCCACA
CCACGAGCTCAACCCCATGCGGGTCC
TGCTAAAGATCGCCAAGTCAGACCCT
CCCACGCTGCTCACGCCCTCCAAGTG
GTCTGTAGAGTTCCGTGACTTCCTGA
AGATAGCCCTGGATAAGAACCCAGAA
ACCCGACCCAGTGCCGCGCAGCTGCT
GGAGCATCCCTTCGTCAGCAGCATCA
CCAGTAACAAGGCTCTGCGGGAGCTG
GTGGCTGAGGCCAAGGCCGAGGTGAT
GGAAGAGTGA
|
Final protein sequence: The following mutations have been detected in that clone with respect to the reference sequence: V62A, E136V, G317E. The mutations have been confirmed by electron density and ESI-MS. smRKSREYEHVRRDLDPNEVWEIVGE
LGDGAFGKVYKAKNKETGALAAAKVI
ETKSEEELEDYIVEIEILATCDHPYI
VKLLGAYYHDGKLWIMIEFCPGGAVD
AIMLELDRGLTEPQIQVVCRQMLEAL
NFLHSKRIIHRDLKAGNVLMTLEGDI
RLADFGVSAKNLKTLQKRDSFIGTPY
WMAPEVVMCETMKDTPYDYKADIWSL
GITLIEMAQIEPPHHELNPMRVLLKI
AKSDPPTLLTPSKWSVEFRDFLKIAL
DKNPETRPSAAQLLEHPFVSSITSNK
ALRELVAEAKAEVMEE |
Host: BL21 (DE3)-R3-pRARE2 |
Growth medium, induction protocol: 4x 10 ml from a 50 ml overnight culture in LB media containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol were used to inoculate 4x 1 L of LB media containing 35 µg/ml kanamycin (no chloramphenicol) in 2 L baffled shaker flasks. Cultures were grown at 37°C in a shaking incubator until the OD600 reached ~0.4. After that the temperature was adjusted to 20°C. Expression was induced using 0.5 mM IPTG at an OD600 of 0.6, and the expression left overnight. The cells were collected by centrifugation and the pellet resuspended in binding buffer and frozen. Binding buffer: 50mM HEPES pH 7.5, 200 mM NaCl, 20 mM imidazole, 0.5 mM TCEP, 0.2 mM PMSF. |
Extraction buffer, extraction method: Cell pellets were lysed by sonication. The lysate was clarified by centrifugation and the supernatant collected for purification. |
Column 1: Ni-affinity chromatography. |
Buffers: Binding buffer: 50 mM HEPES pH 7.4, 200mM NaCl, 20 mM Imidazole, 0.5 mM TCEP. Wash buffer 1: As Binding buffer except 40mM Imidazole. Wash buffer 2: As Binding buffer except 60 mM Imidazole. Elution buffer: As Binding buffer except 250 mM Imidazole. |
Procedure: 10 ml of 50% Ni-Sepharose slurry (GE Healthcare) (5 ml of resin) was applied to a 2 cm diameter gravity column. The column was equilibrated with binding buffer. The lysate was applied to the column which was subsequently washed with binding buffer, wash buffer 1 and 2. STK10 was eluted with 25 ml of elution buffer. The N-terminal His6 tag was cleaved by incubating the protein overnight with TEV protease. |
Column 2: Size exclusion chromatography (Superdex S200, 16/60) |
SEC-Buffers: 20 mM Hepes, pH 7.4, 300 mM NaCl, 0.5 mM TCEP. |
Procedure: The fractions eluted from the Ni-affinity chromatography were concentrated to about 10 ml by ultrafiltration. The concentrated protein was applied to the column (pre-equilibrated in SEC buffer) at a flow rate of 0.7 ml/min. |
Column 3: Ni-affinity chromatography (reverse purification). |
Procedure: Fractions from the gel filtration containing STK10 were pooled and passed through a column of Ni-Sepharose (2 ml, pre-equilibrated in SEC Buffer). The resin was eluted with SEC Buffer containing 10, 20, 30, 40 mM imidazole. STK10 was present in the flow-through and 10 mM imidazole elution. |
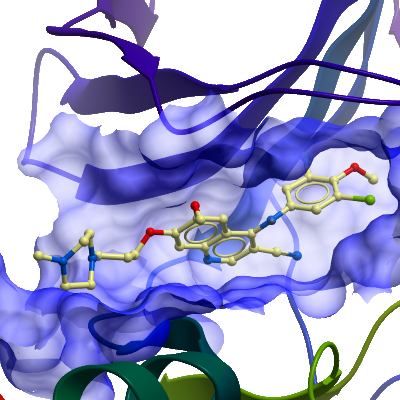
Crystallization: STK10 was concentrated to 13.6 mg/ml. Crystals were obtained using the vapor diffusion method using this protein mixed with 1 mM Bosutinib-like compound, mixed 1:1 with a well solution containing 0.1M SPG pH 7.0, 60% MPD. Crystals appeared after a couple of days at 4°C. |
Data Collection: Crystals were directly flash frozen in liquid nitrogen. Diffraction data were collected at the Diamond beam line I04. |