ACVR1 (also known as ALK2) belongs to the bone morphogenetic protein (BMP) receptor family of transmembrane serine/threonine kinases. Secreted BMPs, which are members of the TGF-beta superfamily involved in endochondral bone formation and embryogenesis, induce heteromeric assembly of type II and type I receptors. Five type II and seven type I receptors, also termed activin-receptor like kinases (ALK1-7) are identified in mammals. All ALKs have a similar sequence and domain structure composed of a ligand-binding extracellular domain, a transmembrane domain, and a cytoplasmic kinase domain (ten Dijke et al., 1993). The type II receptor functions to activate the type I receptor by phosphorylation of its glycine-serine rich (GS) domain. Subsequently, the type I receptor binds and phosphorylates members of the SMAD family of transcription factors. Binding of FKBP12 to the GS domain stabilizes the inactive confirmation of type I receptor and thus prevents leaky activation (Bocciardi et al., 2009; Graham and Peng, 2006).
Mouse models have shown the importance of ACVR1 for cardiac, lens and skeletal development, epithelial-mesenchymal transformation, Müllerian duct regression, generation of primordial gem cells and also for normal mesoderm formation in extraembryonic tissues at the time of gastrulation (Rajagopal et al., 2008; Yu et al., 2008) (Fukuda et al., 2006; Komatsu et al., 2007) (Wang et al., 2005) (de Sousa Lopes et al., 2004; Kaartinen et al., 2004) (Dudas et al., 2004) (Gu et al., 1999) (Mishina et al., 1999) (Ameerun et al., 1996) (Verschueren et al., 1995). Homozygous mutant mice of ACVR1 show morphological defects at E7.0 and have died by E9.5.
A single mutation of 617G-A resulting in an R206 to H conversion in the GS domain of ACVR1 is the primary cause of fibrodysplasia ossificans progressiva (FOP), a rare autosomal dominant disorder of skeletal malformations and progressive extraskeletal ossification (Shore et al., 2006). Affected individuals show heterotopic ossification in childhood, which can be induced by trauma or may occur without warning. Bone formation is episodic and progressive, leading to extra-articular ankylosis of all major joints of the axial and appendicular skeleton, rendering movement impossible. Kinase inhibitors of ACVR1 offer a potential therapeutic against FOP and have shown some efficacy in mice (Yu et al. 2008)).
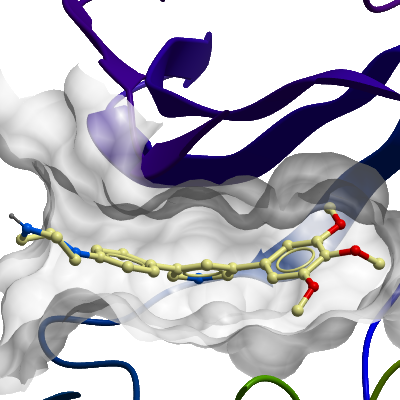
Here we present the structure of the ACVR1 kinase domain in complex with the inhibitor LDN-213844 refined at 2.56 Å resolution.
LDN-213844