Molecular Biology
Entry Clone Accession: IMAGE:2819332
Entry Clone Source: MGC
SGC Construct ID: TSTA3A-c013
Protein Region: S7-K321
Vector: pNIC28-Bsa4
Tag: N-6HIS;N-TEV
Host: BL21(DE3)-R3
Sequence (with tag(s)): MHHHHHHSSGVDLGTENLYFQSMRILVTGGSGLVGKAIQKVVADGAGLPGEDWVFVSSKDADLTDTAQTRALFEKVQPTHVIHLAAMVGGLFRNIKYNLDFWRKNVHMNDNVLHSAFEVGARKVVSCLSTCIFPDKTTYPIDETMIHNGPPHNSNFGYSYAKRMIDVQNRAYFQQYGCTFTAVIPTNVFGPHDNFNIEDGHVLPGLIHKVHLAKSSGSALTVWGTGNPRRQFIYSLDLAQLFIWVLREYNEVEPIILSVGEEDEVSIKEAAEAVVEAMDFHGEVTFDTTKSDGQFKKTASNSKLRTYLPDFRFTPFKQAVKETCAWFTDNYEQARK
Sequence after tag cleavage: SMRILVTGGSGLVGKAIQKVVADGAGLPGEDWVFVSSKDADLTDTAQTRALFEKVQPTHVIHLAAMVGGLFRNIKYNLDFWRKNVHMNDNVLHSAFEVGARKVVSCLSTCIFPDKTTYPIDETMIHNGPPHNSNFGYSYAKRMIDVQNRAYFQQYGCTFTAVIPTNVFGPHDNFNIEDGHVLPGLIHKVHLAKSSGSALTVWGTGNPRRQFIYSLDLAQLFIWVLREYNEVEPIILSVGEEDEVSIKEAAEAVVEAMDFHGEVTFDTTKSDGQFKKTASNSKLRTYLPDFRFTPFKQAVKETCAWFTDNYEQARK
DNA Sequence: CATATGCACCATCATCATCATCATTCTTCTGGTGTAGATCTGGGTACCGAGAACCTGTACTTCCAATCCATGCGGATTCTAGTGACAGGGGGCTCTGGGCTGGTAGGCAAAGCCATCCAGAAGGTGGTAGCAGATGGAGCTGGACTTCCTGGAGAGGACTGGGTGTTTGTCTCCTCTAAAGACGCCGATCTCACGGATACAGCACAGACCCGCGCCCTGTTTGAGAAGGTCCAACCCACACACGTCATCCATCTTGCTGCAATGGTGGGGGGCCTGTTCCGGAATATCAAATACAATTTGGACTTCTGGAGGAAAAACGTGCACATGAACGACAACGTCCTGCACTCGGCCTTTGAGGTGGGCGCCCGCAAGGTGGTGTCCTGCCTGTCCACCTGTATCTTCCCTGACAAGACGACCTACCCGATAGATGAGACCATGATCCACAATGGGCCTCCCCACAACAGCAATTTTGGGTACTCGTATGCCAAGAGGATGATCGACGTGCAGAACAGGGCCTACTTCCAGCAGTACGGCTGCACCTTCACCGCTGTCATCCCCACCAACGTCTTCGGGCCCCACGACAACTTCAACATCGAGGATGGCCACGTGCTGCCTGGCCTCATCCACAAGGTGCACCTGGCCAAGAGCAGCGGCTCGGCCCTGACGGTGTGGGGTACAGGGAATCCGCGGAGGCAGTTCATATACTCGCTGGACCTGGCCCAGCTCTTTATCTGGGTCCTGCGGGAGTACAATGAAGTGGAGCCCATCATCCTCTCCGTGGGCGAGGAAGATGAGGTCTCCATCAAGGAGGCAGCCGAGGCGGTGGTGGAGGCCATGGACTTCCATGGGGAAGTCACCTTTGATACAACCAAGTCGGATGGGCAGTTTAAGAAGACAGCCAGTAACAGCAAGCTGAGGACCTACCTGCCCGACTTCCGGTTCACACCCTTCAAGCAGGCGGTGAAGGAGACCTGTGCTTGGTTCACTGACAACTACGAGCAGGCCCGGAAGTGACAGTAAAGGTGGATACGGATCCGAA
Protein Expression
Medium: LB
Antibiotics: Kanamycin
Procedure: The plasmid was transformed into E. coli, strain BL21 (DE3)-R3-pRARE. For protein expression 5 µl of a glycerol stock were used to inoculate 50 ml LB medium containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol. A starter culture was grown over night at 37°C and used on the following day to inoculate LB medium containing 50 µg/ml kanamycin. Cells were grown at a temperature of 37°C to a OD600 of approx. 1.0 whereupon temperature was reduced to 18°C and cells were induced with 0.2 mM IPTG. Expresion continued overnight at 18oC. Cells were harvested by centrifugation at 4°C at 6500 rpm for 11min. The supernatant was discarded and cell pellets stored at -20°C until further purification.
Protein Purification
Procedure: Cells were slowly thawed on ice, resuspended in lysis buffer (50 mM HEPES pH 7.5, 500 mM NaCl, 5 % (w/v) glycerol, 20 mM imidazole, 0.5 mM TCEP, 1 mM PMSF) and homogenised using an Avestin C-5 fluidizer. Insoluble material was removed by centrifugation (4°C, 16500 rpm, 45min. The supernatant was purified by Nickel affinity (Ni-sepharose; wash buffer: 50 mM Tris-HCl pH 7.5, 500 mM NaCl, 5% glycerol, 30 mM imidazole; elution buffer: 50 mM HEPES pH 7.5, 50 mM NaCl, 5% glycerol, 250 mM imidazole) and size exclusion (Superdex S200; gel filtration buffer: 10 mM HEPES pH 7.5, 500 mM NaCl, 5% glycerol, 1mM PMSF, 0.5 mM TCEP) chromatography. Protein was concentrated to 12 to 15 mg/ml and stored at -80oC.
Mass-spec Verification: Yes
Structure Determination
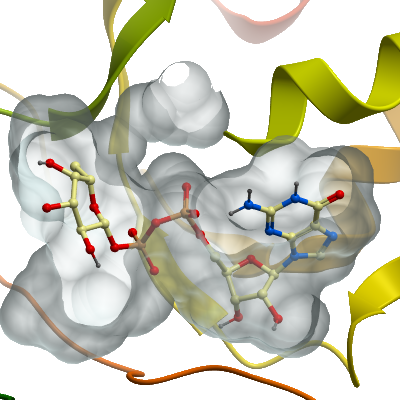
Crystallization: Crystals were grown by vapour diffusion in sitting drop at 20°C by setting up 12.6 mg/ml of protein in the presence of 5 mM NADP+ and 10 mM GDP-L-fucose. Pyramidal crystals appeared in a sitting drop consisting of 75 nl protein and 75 nl well solution which had been equilibrated against 20 µl well solution containing 25% PEG 3350, 0.1 M bis-tris-propane pH 7.5, 0.15 M NaCl. Crystals were mounted in the presence of 25% PEG 400 and flash cooled in liquid nitrogen.
Data Collection: Beamline: Dmnd I04; Resolution: 2.6 Å
Data Processing: After processing with XDS and further editing with Aimless phases were derived in P1 using chain B from PDB ID 4B8Z as search model running Phaser within the CCP4 Suite. A total of twelve molecules were found forming six dimers. Subsequent model building was carried out in Coot and for refinement Refmac5 was used.