Protein methyltransferases play diverse roles in various biological pathways mainly through
site-specific methylation of lysines/arginines in the target proteins. All the protein arginine
methyltransferases are part of the seven-β-strand family, while most of the protein lysine
methyltransferases belong to the SET-domain containing protein family. Recently, a new group
of protein methyltransferase has been identified through genome wide analysis. This group
of protein methyltransferases shows distant homology with PRMTs. They all belong to the
seven-β-strand family of methyltransferases and contain a Rossmann fold as their cofactor and
substrate binding site. Biochemical studies have shown that these proteins are lysine-specific
methyltransferases, with a preference for molecular chaperons as their substrates.
Human methyltransferase-like protein 21A (METTL21A) is a member of the novel PMT family.
METTL21A has been shown to trimethylates a conserved Lys residue found in human Hsp70s.
Methylation of Hsp70s by METTL21A does not have an effect on HSP70 ATPase activity, but
does affect its ability to bind to α-synuclein, whose aggregation is associated with Parkinson's
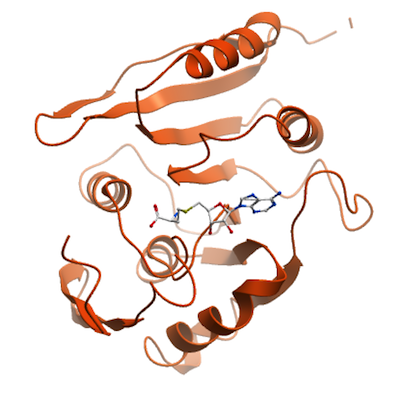
disease. We have solved the crystal structure of human METTL21A bound with S-adenosyl-L-
homocysteine at 2.28 Å resolution.