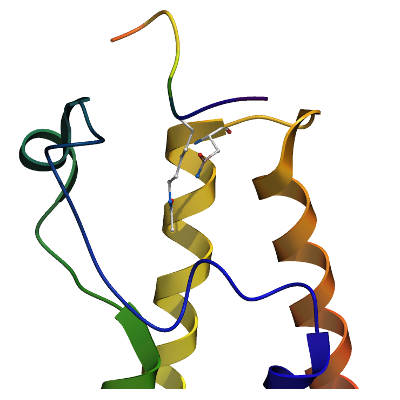
BAZ2A
PDB:4QBM
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:BAZ2A_HUMAN Q9UIF9
Entry Clone Source:synthetic
SGC Clone Accession:
Tag:mhhhhhhssgvdlgtenlyfq*s(m) TEV-cleavable (*) N-terminal his6 tag.
Host:BL21(DE3)-R3-pRARE2
Construct
Prelude:
Sequence:SMHSDLTFCEIILMEMESHDAAWPFLEPVNPRLVSGYRRIIKNPMDFSTMRERLLRGGYTSSEEFAADALLVFDNCQTFNEDDSEVGKAGHIMRRFFESRWEEFYQ
Vector:pNIC28-Bsa4
Growth
Medium:
Antibiotics:
Procedure:Best expression construct BAZ2A-c002 (KanR) bromodomain was transformed into Escherichia coli competent BL21(DE3) Rosetta (ChlR) cells or into BL21 (DE3)-R3-pRARE2 cells (phage-resistant derivative with a pRARE plasmid encoding rare codon tRNAs). Cells were grown at 37°C in Terrific Broth from overnight cultures until A600 reached between 0.6-0.8, then the media was cooled and 0.2 mM isopropyl-β-Dthiogalactopyranoside (IPTG) was added to induce the protein expression at 20°C for 16 hours
Purification
Procedure
STEP1: after centrifugation, the supernatant was loaded onto the nickel column and eluted in an imidazole linear gradient. Binding buffer 20 mM HEPES pH 7.5; 500 mM NaCl; 10 mM imidazole, 0.5 mM TCEP; Elution buffer 20 mM HEPES pH 7.5; 500 mM NaCl; 500 mM imidazole and 0.5 mM TCEP.STEP2: the eluted protein was collected and treated overnight with TEV (Tobacco Etch Virus) protease at 4ºC to remove the N terminal tag. Dialysis buffer 20 mM Hepes pH 7.5, 500 mM NaCl, 0.5 mM TCEP (no imidazole)STEP3: digested protein was loaded onto a nickel column again to remove the cleaved 6His tag and the hexa-histidine expression TEV protease used. The flow through containing the untagged protein was collected and further purified. Binding buffer 20 mM Hepes pH 7.5, 500 mM NaCl, 0.5 mM TCEP, 20 mM imidazole; Elution buffer 20 mM Hepes pH 7.5, 500 mM NaCl, 0.5 mM TCEP, 500 mM ImidazoleSTEP4: a size exclusion chromatography (HiLoad 16/600 Superdex 75) is performed to remove soluble aggregates, buffer 20 mM Hepes pH 7.5, 500 mM NaCl, 2 mM DTT.
Extraction
Procedure
Frozen pellets were thawed and resuspended in binding buffer containing 20 mM HEPES pH 7.5; 500 mM NaCl; 10 mM imidazole and 0.5 mM TCEP. Cells were lysed using an EmulsiFlex-C5 high-pressure homogenizer (Avestin - Mannheim, Germany) or high-pressure cell disruption (Constant Systems Limited) in loading buffer A from the nickel affinity column (HisTrap Chelating FF 5ml) in the presence of Protease Inhibitor Cocktail EDTA-free Lysates were cleared by centrifugation (14,000 x g for 45h at 4ºC, JLA 16.250 rotor, on a Beckman Coulter Avanti J-20 XP centrifuge).
Concentration:The cleaved purified protein buffer exchange to 20 mM Hepes pH 7.5, 500 mM NaCl, 2 mM DTT and concentrated in a VivaSpin (3 K MWCO) to 23 mg/ml prior to crystallization trials. The protein concentration was determined spectrophotometrically.
Ligand
MassSpec:Observed mass without histidine tag 12495.098, (calculated mass without histidine tag, 12495.0).
Crystallization:Crystallization of Bromodomain of BAZ2A in the free-form: clusters of thin needle-shaped crystals of the free-form of single BAZ2A bromodomain were obtained by mixing equal volumes of 23 mg/ml and crystallization buffer (0.1 M Tris-HCl pH 8.4, 0.25 M MgCl2, 22% PEG 3350) at 4ºC. Several rounds of macro-seeding were needed to obtain thicker and isolated bar-shaped crystals using the sitting drop vapour diffusion method. The protein buffer was 20 mM Hepes pH 7.5, 200 mM NaCl, 2 mM DTT. A single crystal was flash-frozen at 100 K in a nitrogen gas stream in the cryoprotectant with 15% ethylene glycol.Histone peptide H4K16acK20ac complex crystallization with TIP5 bromodomain: protein was concentrated up to 17 mg/ml and co-crystallised with 1:5 M excess of the 9-mer peptide in 150 nl + 150 nl + 20 nl seeds (from 2nd round) sitting drop at 4ºC. Prism-like crystals appeared in 2-3 days and the seeding helped to prevent twinning issues. The optimised crystallisation conditions were 0.1 M sodium phosphate monobasic, 0.1 M potassium phosphate monobasic, 0.1M MES pH 6.5, 2 M sodium chloride. 20% glycerol was used as the cryoprotectant
NMR Spectroscopy:
Data Collection:Crystallization of Bromodomain of BAZ2A in the free-form: clusters of thin needle-shaped crystals of the free-form of single BAZ2A bromodomain were obtained by mixing equal volumes of 23 mg/ml and crystallization buffer (0.1 M Tris-HCl pH 8.4, 0.25 M MgCl2, 22% PEG 3350) at 4°C. Several rounds of macro-seeding were needed to obtain thicker and isolated bar-shaped crystals using the sitting drop vapour diffusion method. The protein buffer was 20 mM Hepes pH 7.5, 200 mM NaCl, 2 mM DTT. A single crystal was flash-frozen at 100 K in a nitrogen gas stream in the cryoprotectant with 15% ethylene glycol.Histone peptide H4K16acK20ac complex crystallization with TIP5 bromodomain: protein was concentrated up to 17 mg/ml and co-crystallised with 1:5 M excess of the 9-mer peptide in 150 nl + 150 nl + 20 nl seeds (from 2nd round) sitting drop at 4°C. Prism-like crystals appeared in 2-3 days and the seeding helped to prevent twinning issues. The optimised crystallisation conditions were 0.1 M sodium phosphate monobasic, 0.1 M potassium phosphate monobasic, 0.1M MES pH 6.5, 2 M sodium chloride. 20% glycerol was used as the cryoprotectant
Data Processing: