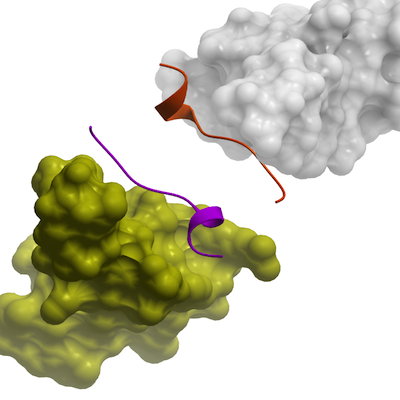
In the complex structure, the PPCY peptide had the sequence N-TPEAPPCYMDVI-C, corresponding to residues 327′-338′ of TXNIP (hereafter, TXNIP residues numbers are marked with a prime (′) to distinguish them from the Itch residue numbers). The peptide's P331' and P332' form a polyproline type II helix, whereas residues 334′-337′ adopt a helical fold. The binding site of the peptide is located on one face of the WW sheet and consists of two canonical grooves, namely the XP groove and tyrosine-binding groove (Tyr groove). P331′ and P332′ insert into the XP groove by stacking against the conserved W466 and Y455 residues of WW3, respectively. The side chain of Y334′ is accommodated in the Tyr groove formed by the conserved residues V457, H459 and R462 of WW3. The side chain of V337′ presumably points toward the hydrophobic pocket formed by the side chain of Y334′ in PPCY peptide and V457, T464 in WW3 domains, but was poorly resolved by electron density. In addition to the hydrophobic packing, some hydrogen bonds between the peptide and WW domains contribute to the stabilization of the complex, for example between the carbonyl of P332′ and the hydroxyl of T464, along with the phenolic hydroxyl of Y334′ and the imidazole Nδ of H459.