|
Entry Clone Source: strong>Gregg Morin, University of British Columbia
|
|
Entry Clone Accession:
|
|
SGC Construct ID: CRK7A-c021
|
|
Amplified DNA sequence:
TACTTCCAATCCATGACAGAAAGCGACTGGGGGAAACGCTGTGTGGACAAGTTTGACATTATTGGGATTATTGGAGAAGGAACCTA
TGGCCAAGTATATAAAGCCAAGGACAAAGACACAGGAGAACTAGTGGCTCTGAAGAAGGTGAGACTAGACAATGAGAAAGAGGGCT
TCCCAATCACAGCCATTCGTGAAATCAAAATCCTTCGTCAGTTAATCCACCGAAGTGTTGTTAACATGAAGGAAATTGTCACAGAT
AAACAAGATGCACTGGATTTCAAGAAGGACAAAGGTGCCTTTTACCTTGTATTTGAGTATATGGACCATGACTTAATGGGACTGCT
AGAATCTGGTTTGGTGCACTTTTCTGAGGACCATATCAAGTCGTTCATGAAACAGCTAATGGAAGGATTGGAATACTGTCACAAAA
AGAATTTCCTGCATCGGGATATTAAGTGTTCTAACATTTTGCTGAATAACAGTGGGCAAATCAAACTAGCAGATTTTGGACTTGCT
CGGCTCTATAACTCTGAAGAGAGTCGCCCTTACACAAACAAAGTCATTACTTTGTGGTACCGACCTCCAGAACTACTGCTAGGAGA
GGAACGTTACACACCAGCCATAGATGTTTGGAGCTGTGGATGTATTCTTGGGGAACTATTCACAAAGAAGCCTATTTTTCAAGCCA
ATCTGGAACTGGCTCAGCTAGAACTGATCAGCCGACTTTGTGGTAGCCCTTGTCCAGCTGTGTGGCCTGATGTTATCAAACTGCCC
TACTTCAACACCATGAAACCGAAGAAGCAATATCGAAGGCGTCTACGAGAAGAATTCTCTTTCATTCCTTCTGCAGCACTTGATTT
ATTGGACCACATGCTGACACTAGATCCTAGTAAGCGGTGCACAGCTGAACAGACCCTACAGAGCGACTTCCTTAAAGATGTCGAAC
TCAGCAAAATGGCTCCTCCAGACCTCCCCCACTGGCAGGATTGACAGTAAAGGTGGATA
|
|
Expressed protein sequence:
MGHHHHHHSSGVDLGTENLYFQSMTESDWGKRCVDKFDIIGIIGEGTYGQVYKAKDKDTGELVALKKVRLDNEKEGFPITAIREI
KILRQLIHRSVVNMKEIVTDKQDALDFKKDKGAFYLVFEYMDHDLMGLLESGLVHFSEDHIKSFMKQLMEGLEYCHKKNFLHRDI
KCSNILLNNSGQIKLADFGLARLYNSEESRPYTNKVITLWYRPPELLLGEERYTPAIDVWSCGCILGELFTKKPIFQANLELAQL
ELISRLCGSPCPAVWPDVIKLPYFNTMKPKKQYRRRLREEFSFIPSAALDLLDHMLTLDPSKRCTAEQTLQSDFLKDVELSKMAP
PDLPHWQD
|
|
Vector: pFB-LIC-Bse
|
|
Tags and additions: MGHHHHHHSSGVDLGTENLYFQ*SM. cleavable N-terminal
hexahistidine tag.
|
|
Cyclin K
|
|
Entry Clone Source: MGC
|
|
Entry Clone Accession: BC015935
|
|
SGC Construct ID: CCNKA-c001
|
|
Amplified DNA sequence:
TACTTCCAATCCATGTCAGTAACTTCAGCAAACCTGGACCACACAAAGCCATGTTGGTACTGGGATAAGAAAGACTTGGCT
CATACACCCTCACAACTTGAAGGACTTGATCCAGCCACCGAGGCCCGGTACCGCCGAGAGGGCGCTCGGTTCATCTTTGAT
GTGGGCACACGTTTGGGGCTACACTATGATACCCTGGCAACTGGAATAATTTATTTTCATCGCTTCTATATGTTTCATTCC
TTCAAGCAATTCCCAAGATATGTGACAGGAGCCTGTTGCCTCTTTCTGGCTGGGAAAGTAGAAGAAACACCAAAAAAATGT
AAAGATATCATCAAAACAGCTCGTAGTTTATTAAATGATGTACAATTTGGCCAGTTTGGAGATGACCCAAAGGAGGAAGTA
ATGGTTCTGGAGAGAATCTTACTGCAGACCATCAAGTTTGATTTACAGGTAGAACATCCATACCAGTTCCTACTAAAATAT
GCAAAGCAACTCAAAGGTGATAAAAACAAAATTCAAAAGTTGGTTCAAATGGCATGGACATTTGTAAATGACAGTCTCTGC
ACCACCTTGTCACTGCAGTGGGAACCAGAGATCATAGCAGTAGCAGTGATGTATCTCGCAGGACGTTTGTGCAAATTTGAA
ATACAAGAATGGACCTCCAAACCCATGTATAGGAGATGGTGGGAGCAGTTTGTTCAAGATGTCCCGGTCGACGTTTTGGAA
GACATCTGCCACCAAATCCTGGATCTTTACTCACAAGGAAAACAACAGATGCCTCATTGACAGTAAAGGTGGATA
|
|
Expressed protein sequence:
MGHHHHHHSSGVDLGTENLYFQSMSVTSANLDHTKPCWYWDKKDLAHTPSQLEGLDPATEARYRREGARFIFDVGTRLGL
HYDTLATGIIYFHRFYMFHSFKQFPRYVTGACCLFLAGKVEETPKKCKDIIKTARSLLNDVQFGQFGDDPKEEVMVLERI
LLQTIKFDLQVEHPYQFLLKYAKQLKGDKNKIQKLVQMAWTFVNDSLCTTLSLQWEPEIIAVAVMYLAGRLCKFEIQEWT
SKPMYRRWWEQFVQDVPVDVLEDICHQILDLYSQGKQQMPH
|
|
Vector:pFB-LIC-Bse
|
|
Tags and additions: MGHHHHHHSSGVDLGTENLYFQ*SM. cleavable
N-terminal hexahistidine tag.
|
|
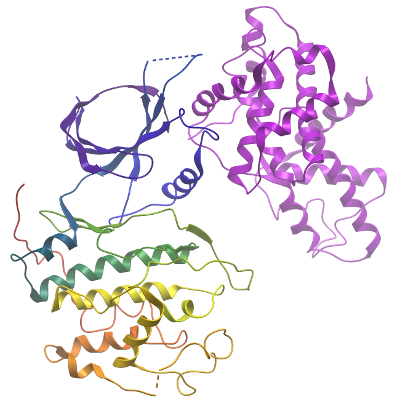
CDK12/cyclin K Complex
|
|
Host: SF9 Spodoptera frugiperda Insect cells
|
|
Material and Methods
|
|
Co-expression of CDK12 and CCNK:
Sf9 cells were grown in Insect-Xpress media (Lonza), to a density of 2x106cells/ml
and were infected with recombinant CDK12 and CCNK baculovirus (P2 virus stocks;
1.5 ml of CDK12 virus stock and 1.5 ml of CCNK virus stock, per 1L of cell culture).
Cells were shaken at 95 rpm at 27°C in an Innova shaker. After 72 hours post-infection
the cultures were harvested by centrifugation for 25min at 900xg at 4°C. Cell pellet
from 1L flasks were made up to 50 ml in binding buffer (50 mM Hepes, pH 7.5;
500 mM NaCl; 5% Glycerol; 5 mM imidazole), transferred to 50 ml falcon tubes,
and stored at -20°C. Calbiochem protease inhibitor cocktail set III was added
to the cell suspension at a 1:5000 dilution.
|
|
Extraction method:
The frozen cells were thawed and lysed
by ultrasonication (Sonic, Vibra Cell) over 12 min at 35% amplitude, with the
sonicator pulsing ON for 5 sec and OFF for 10 sec. Polyethylenimine (PEI) was
added to a final concentration of 0.5% to precipitate DNA and the cell lysate
clarified by centrifugation at 21,000 RPM for 1 hour at 4°C. The supernatant was
recovered for purification.
|
|
Column 1:
Ni-Affinity Chromatography. 5 ml of 50 % nickel-sepharose resin slurry (GE Healthcare)
was applied onto a 1.5 x 10 cm column. The column was washed with ultra-pure water,
then pre-equilibrated with binding buffer.
|
|
Buffers:
Binding Buffer: 500mM NaCl, 50mM HEPES pH 7.5, 5% Glycerol, 5mM Imidazole, 0.5mM TCEP
Wash Buffer: 500mM NaCl, 50mM HEPES pH 7.5, 5% Glycerol, 30mM Imidazole, 0.5mM TCEP
Elution Buffer I: 500mM NaCl, 50mM HEPES pH 7.5, 5% Glycerol, 50mM Imidazole, 0.5mM TCEP
Elution Buffer II: 500mM NaCl, 50mM HEPES pH 7.5, 5% Glycerol, 100mM Imidazole, 0.5mM TCEP
Elution Buffer III: 500mM NaCl, 50mM HEPES pH 7.5, 5% Glycerol, 150mM Imidazole, 0.5mM TCEP
Elution Buffer IV: 500mM NaCl, 50mM HEPES pH 7.5, 5% Glycerol, 250mM Imidazole, 0.5mM TCEP
Immobilised metal affinity chromatography procedure:
The supernatant, following centrifugation, was filtered and applied by gravity flow onto the
Ni-sepharose column. The bound protein was then washed with 100 ml binding buffer and subsequently
with 60 ml wash buffer. CDK12/CCNK protein was then eluted by applying a step gradient of imidazole
- using 10 ml fractions of elution buffer with increasing concentration of imidazole (50 mM, 100mM,
150mM and 250 mM). Elution fractions were analyzed by SDS PAGE and the 250 mM imidazole fraction
was kept for subsequent steps. 10 mM DTT was added for overnight storage at 4?C.
Enzymatic treatment: 0.1mg of TEV protease was added to the Ni-eluted protein to remove the tag.
Incubation was overnight at 4°C
Phosphorylation of CDK12 by Candida albicans, Cak1 (CaCAK):
Following tag cleavage, the protein complex was concentrated to 1.1 ml using an Amicon Ultra-15
filter with a 10 kDa cut-off. The complex was mixed with Candida albicans, Cak1 (CaCAK) (0.07mg/ml)
at room temperature, in the presence of 1 mM ATP, 5 mM MgCl2 and 0.1 mM MnCl2 until CDK12
was singularly phosphorylated as monitored by ESI-MS. The protein was purified further by size
exclusion chromatography.
|
|
Column 2: Size Exclusion Chromatography - S75 HiLoad 26/60 Superdex
column (GE Healthcare) run on ÄKTA-Express
|
|
Buffer:
Gel Filtration buffer: 300 mM NaCl, 50 mM Hepes pH 7.5, 0.5mM TCEP
Gel filtration procedure: Prior to applying the protein, the S75 HiLoad 26/60 Superdex column was
washed and equilibrated with gel filtration buffer. The concentrated protein was diluted in gel
filtration buffer, to around 3ml and directly applied onto the equilibrated S75 HiLoad 26/60 Superdex
column, and run at a flow-rate of 1 ml/min. Fractions (1.8 ml each) containing the protein were
pooled together. The eluted protein was supplemented with 5 mM L-arginine, 5 mM L-glutamate and 5 mM
dithiothreitol (DTT).
|
|
Column 3:
Reverse Ni-Affinity Chromatography. 0.5 ml of 50 %
nickel-sepharose resin slurry (GE Healthcare) was applied onto a Bio-Rad Poly-Prep
drip column. The column was washed with ultra-pure water, then pre-equilibrated with
gel filtration buffer.
|
|
Buffer:
Gel Filtration buffer: 300 mM NaCl, 50 mM Hepes pH 7.5, 0.5mM TCEP
Binding Buffer: 500mM NaCl, 50mM HEPES pH 7.5, 5% Glycerol, 5mM Imidazole, 0.5mM TCEP
Wash Buffer: 500mM NaCl, 50mM HEPES pH 7.5, 5% Glycerol, 30mM Imidazole, 0.5mM TCEP
Elution Buffer IV: 500mM NaCl, 50mM HEPES pH 7.5, 5% Glycerol, 250mM Imidazole, 0.5mM TCEP
Reverse Ni-Affinity Chromatography:
Protein-containing fractions were pooled and reverse nickel-affinity purification applied to further
purify the protein and remove His-tagged CaCAK, prior to concentrating for crystallization.
The concentrated sample from gel filtration was applied by gravity flow onto the Ni-sepharose column.
The column was then washed sequentially with 10 ml gel filtration buffer, 10 ml binding buffer,
10 ml wash buffer and 10 ml elution buffer (containing 250mM imidazole). Fractions were analyzed
by SDS PAGE and showed the CDK12 complex has eluted in the binding buffer fraction. 10 mM DTT was
added to the protein and the sample was concentrated for crystallization trials.
|
|
Mass spec characterization:
The intact mass of the protein was
confirmed by Electrospray Ionisation/Time-of-Flight Mass Spectrometry (ESI-MS, Agilent
Technologies). The purified protein complex had an experimental mass of 37.685 and
30.403 kDa, as expected from primary sequences of CDK12 and CCNK, respectively. Following
CAK treatment the CDK12 mass shifted to 37.768 kDa consistent with a single phosphorylation.
Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase
HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser.
Proteins were desalted prior to mass spectrometry by rapid elution off a C3 column with
a gradient of 5-95% methanol in water with 0.1% formic acid.
|
|
Crystallisation of the CDK12/CCNK complex:
Protein was buffered in
500mM NaCl, 50mM HEPES pH 7.5, 5% Glycerol, 5mM Imidazole, 0.5mM TCEP, 10 mM DTT and
concentrated to 4.1 mg/mL (calculated using a complex MW of 68071 Da and an extinction
co-efficient of 98780 M-1 cm-1). Subsequently, 1mM AMPPNP was added to the concentrated
protein. For crystallisation trials, 1mM MgCl2 was added to the mother liquor reservoir
of each condition prior to plating. Crystals were grown at 4°C in 150 nl sitting drops mixing
100 nl protein solution with 50 nl of a reservoir solution comprising 20% PEG3350,
10% ethylene glycol, 0.1M Bis-tris propane pH 6.5, 0.2M sodium nitrate, 1mM MgCl2.
Before mounting, crystals were cryo-protected with mother liquor supplemented with an
additional 15% ethylene glycol and vitrified in liquid nitrogen.
|
|
Data Collection: Resolution:3.15 Å resolution
X-ray source:
Diffraction data were collected at 100 K on Diamond Light Source beamline I24, using
monochromatic radiation at wavelength 0.9686 Å
|