DPF3
PDB:5I3L
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:NP_001267471.1.
Entry Clone Source:JMC01F:A01
SGC Clone Accession:DPF3 (JMC01J-D05): K250-K368
Tag:N-terminal tag: MHHHHHHSSGRENLYFQG
Host:Escherichiacoli BL21 (DE3)
Construct
Prelude:
Sequence:MHHHHHHSSGRENLYFQGKGPDGTVIPNNYCDFCLGGSNMNKKSGRPEELVSCADCGRSGHPTCLQFTLNMTEAVKTYKWQCIECKSCILCGTSENDDQLLFCDDCDRGYHMYCLNPPVAEPPEGSWSCHLCWELLK
Vector:pET28-MHL
Growth
Medium:
Antibiotics:
Procedure:Lex system, 37 °C 3-4h, 15 °C 18-24h
Purification
Buffers
Wash buffer: 20 mM Tris pH 7.5, 1 M NaCl, 25 mM imidazole - Elution buffer: 20 mM Tris pH 7.5, 250 mM NaCl, 250 mM imidazole - Gel filtration buffer: 20 mM Tris, pH 7.5, 150 mM NaCl, 1 mM DTT, 50 uM ZnCl2
Procedure
IMAC: Unclarified lysate was mixed with 3-4 mL of Ni-NTA superflow Resin (Qiagen) per 200 mL lysate. The mixture was incubated with mixing for at least 30 minutes at 4°C. The mixture was then loaded onto an empty comLum (BioRad) and washed with 40 mL wash buffer. Samples were eluted from the resin by exposure to 2-3 column volumes (approx. 10-15 mL) of elution buffer. Concentration of eluted protein was estimated by OD280. The eluted protein was treated by TEV to remove the His-tag by dialysis against dialysis buffer containing 20 mM Tris pH 7.5, 150 mM NaCl, 1 mM DTT, 50 uM ZnCl2.Gel filtration chromatography: An XK 16x60 column (GE Healthcare) packed with HighLoad Superdex 75 resin (GE Healthcare) was pre-equilibrated with gel filtration buffer for 1.5 column volumes using an AKTA explorer (GE Healthcare) at a flow rate of 1.0 mL/min. The concentrate sample (approx. 3 mL, concentrated by using 15 mL concentrators with a 3,000 molecular weight cut-off (Amicon Ultra-15, UFC900524, Millipore) at 3750 rpm) from the dialysis step was loaded onto the column at 1 mL/min, and 2mL fractions were collected into 96-well plates (VWR 40002-012) using peak fractionation protocols. Fractions observed by a UV absorption chromatogram to contain the protein were pooled.
Extraction
Buffers
Lysis buffer: 20 mM Tris pH 7.5 (R.T.), 400 mM NaCl
Procedure
Frozen cell pellet contained in bags (Beckman 369256) obtained from 2L of culture were thawed by soaking in warm water. Each cell pellet was resuspended in 200 mL lysis buffer and homogenized using an Ultra-Turrax T8 homogenizer (IKA Works) at maximal setting for 30-60 seconds per pellet. Cell lysis was accomplished by sonication (Virtis408912, Virsonic) on ice: the sonication protocol was 10 seconds pulse at 80% of maximal frequency (8.0), 10 seconds rest, for 10 minutes total sonication time per pellet.
Concentration:Purified proteins were concentrated using 15 mL concentrators with a 3,000 molecular weight cut-off (Amicon Ultra-15, UFC900524, Millipore) at 3750 rpm, typically resulting in a final concentration around 5 mg/mL.
Ligand
MassSpec:
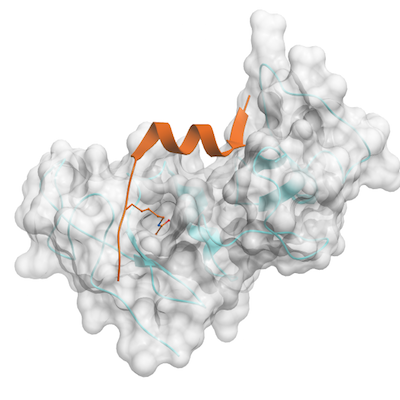
Crystallization:Recombinant human DPF3 tandem PHD domain was mixed with H3K14ac peptide at molar ratio of 1:2 and co-crystallized using the sitting drop vapour diffusion method at 18 °C. The crystals were obtained in a buffer containing 1.4 M NaCitrate, 0.1 M Hepes, pH 7.5, 5% MPD. Crystals were soaked in a cryoprotectant consisting of 100% reservoir solution and 15% glycerol.
NMR Spectroscopy:
Data Collection:
Data Processing: