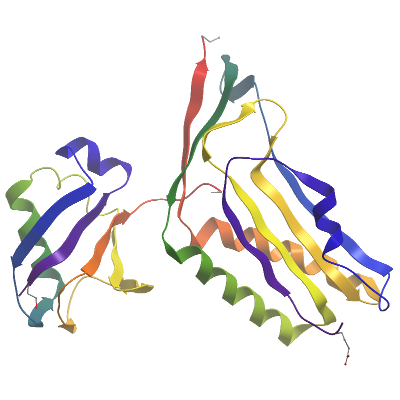
Molecular Biology
Entry Clone Accession:
Entry Clone Source: Complex
SGC Construct ID: XX02MOCS2A-c001
Protein Region: None-None
Vector: Note this is a multibac plasmid generated from a donor and acceptor vector.
Tag: N-6HIS;N-TEV
Host: DH10Bac
Sequence (with tag(s)):
MOCS2A (N-terminal His-tag, TEV-cleavage)
MGHHHHHHSSGVDLGTENLYFQSMVEVLYFAKSAEITGVRSETISVPQEIKALQLWKEIETRHPGLADVRNQIIFAVRQEYVELGDQLLVLQPGDEIAVIPPISGG
MOCS2B
MSAFEPSRKDMDEVEEKSKDVINFTAEKLSVDEVSQLVISPLCGAISLFVGTTRNNFEGKKVISLEYEAYLPMAENEVRKICSDIRQKWPVKHIAVFHRLGLVPVSEASIIIAVSSAHRAASLEAVSYAIDTLKAKVPIWKKEIYEESSTWKGN
Protein Expression
Medium: InsectXpress medium (Lonza)
Antibiotics: Ampicillin
Procedure: protein was expressed in Sf9 cells using 3ml of P2 virus/L cultivated in InsectXpress medium (Lonza) for 72 hours at 27°C.
Protein Purification
Procedure:
Extraction Buffer: 500 mM NaCl, 50 mM HEPES pH 7.5, 20 mM imidazole, 0.5 mM TCEP, 5% glycerol, 1:1000 of Merck Protease Cocktail II.
Binding Buffer: 500 mM NaCl, 50 mM HEPES pH 7.5, 20 mM imidazole, 0.5 mM TCEP, 5% glycerol.
Washing Buffer: 500 mM NaCl, 50 mM HEPES pH 7.5, 40 mM imidazole, 0.5 mM TCEP, 5% glycerol.
Elution Buffer: 500 mM NaCl, 50 mM HEPES pH 7.5, 250 mM imidazole, 0.5 mM TCEP, 5% glycerol.
GF buffer: 500 mM NaCl, 50 mM HEPES pH 7.5, 0.5 mM TCEP, 5% glycerol
A 7L cell pellet was re-suspended in 250ml extraction buffer. Cells were homogenised using a C5 homogenizer at 20Kpsi. The cell debris was removed via centrifugation using a JA 17.00 rotor at 35K for 1hr at 4°C. The supernatant was incubated with 2.5 ml Ni-NTA for 1 hr. Ni-NTA resin bound to the target proteins was washed two times with 25 ml of binding buffer followed by two times with 25 ml of wash buffer and eluted in 5 ml fractions with elution buffer. Elution’s containing protein were loaded onto a S75 gel filtration column and purified via size exclusion. The protein was subsequently incubated with TEV protease at a ratio of 20:1 mg/ml (protein:TEV) overnight at 4°C. Cleaved protein was passed back over 1 ml of Ni-NTA and the flow through containing protein was concentrated to 23.3 mg/ml.
Columns: Column 1: 2.5mL Ni NTA resin; Column 2: S75 Gel Filtration; Column 3: Ni-Rebind;
Concentration: 23.3 mg/ml
Structure Determination
Crystallization: Crystals were grown by vapour diffusion method. Sitting drops containing 100 nL protein (23.3 mg/mL) and 50 nL well solution containing 12% PEG3350, 0.1M potassium nitrate and 0.05M potassium thiocyanate were equilibrated at 20°C.
Data Collection: Beamline: Dmnd I03; Resolution: 2.43 Å
Data Processing: Data was processed using Xia2 auto processing pipeline. The protein crystalised in the hexagonal P65 space group and contained 2 copies of each subunit in the asymmetric unit. The structure was solved via molecular replacement with Phaser. The final model was produced by iterative cycles of restrained refinement and model building using REFMAC5 and COOT