EHMT2
PDB:5TTF
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:GI:18375637
Entry Clone Source:MGC
SGC Clone Accession:
Tag:N-terminal: His-tag with integrated thrombin protease site: MGSSHHHHHHSSGLVPRGS
Host:E.coli BL21 (DE3) codon plus RIL (Stratagen)
Construct
Prelude:
Sequence:gsNRAIRTEKIICRDVARGYENVPIPCVNGVDGEPCPEDYKYISENCETSTMNIDRNITHLQHCTCVDDCSSSNCLCGQLSIRCWYDKDGRLLQEFNKIEPPLIFECNQACSCWRNCKNRVVQSGIKVRLQLYRTAKMGWGVRALQTIPQGTFICEYVGELISDAEADVREDDSYLFDLDNKDGEVYCIDARYYGNISRFINHLCDPNIIPVRVFMLHQDLRFPRIAFFSSRDIRTGEELGFDYGDRFWDIKSKYFTCQCGSEKCKHSAEAIALEQSRLARLD
Vector:p28a-LIC
Growth
Medium:Terrific Broth (TB)
Antibiotics:50 µg/mL of kanamycin
Procedure:EHMT2 was expressed in E.coli BL21 (DE3) codon plus RIL in Terrific Broth (TB) in the presence of 50 µg/mL of kanamycin. Cells were grown at 37°C to OD600 of 1.5 and induced by isopropyl-1-thio-D-galactopyranoside (IPTG), final concentration 1 mM, and incubated overnight at 15°C.
Purification
Buffers
Procedure
The crude extract was cleared by centrifugation and loaded onto 5 mL HiTrap Chelating column (Amersham Biosciences), charged with Ni2+. The column was washed with 10 CV of 20 mM Tris-HCl buffer, pH 8.0, containing 250 mM NaCl, 50 mM imidazole and 5% glycerol, and the protein was eluted with elution buffer (20 mM Tris-HCl, pH 8.0, 250 mM NaCl, 250 mM imidazole, 5% glycerol). The protein was loaded on Superdex200 column (26x60) (Amersham Biosciences), equilibrated with 20 mM Tris-HCl buffer, pH 8.0, and 150 mM NaCl, at flow rate 4 mL/min. Thrombin (Sigma) was added to combined fractions containing EHMT2 and incubated overnight at 4°C. The protein was further purified to homogeneity by ion-exchange chromatography on Source 30Q column (10x10) (Amersham Biosciences), equilibrated with buffer 20 mM Tris-HCl, pH 8.0, and eluted with linear gradient of NaCl up to 500 mM concentration (20CV). Purification yield was 3 mg of the protein per 1L of culture.
Extraction
Buffers
Procedure
Cells were harvested by centrifugation at 7,000 rpm. The cell pellets were frozen in liquid nitrogen and stored at -80°C. For purification the cell paste was thawed and resuspended in lysis buffer (phosphate-buffered saline, pH 7.4, 0.25 M NaCl, 5 mM imidazol, 2 mM ß-mercaptoethanol, 5% glycerol) with protease inhibitor (0.1mM phenylmethyl sulfonyl fluoride, PMSF). The cells were lysed by passing through Microfluidizer (Microfluidics Corp.) at 20,000 psi.
Concentration:10 mg/ml
Ligand
MassSpec:expected MW = 32564.8 Da, measured MW = 32563.41 Da.
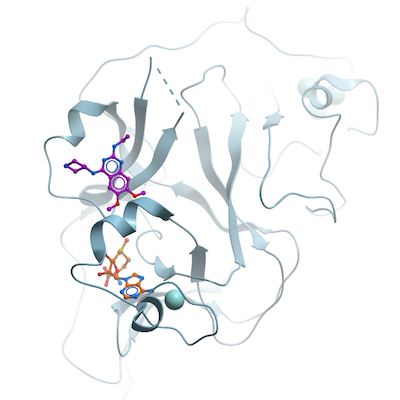
Crystallization:Purified G9a was complexed with S-adenosyl-L-homocysteine (SAH) (Sigma) and MS012 at 1:5:5 molar ratio of protein:SAH:MS012 and crystallized using the sitting drop vapor diffusion method at 20 °C by mixing 0.5 µl of the protein solution with 0.5 µl of the reservoir solution containing 25% PEG 3350, 0.2M NaCl, 0.1 M Bis-Tris propane, pH 6.5.
NMR Spectroscopy:
Data Collection:
Data Processing: