EED
PDB:5TTW
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:BC068995
Entry Clone Source:AU110-F3
SGC Clone Accession:APC049-D10
Tag:N-terminal His6-tag, removed
Host:BL21(DE3)CodonPlus RIL
Construct
Prelude:
Sequence:gKKCKYSFKCVNSLKEDHNQPLFGVQFNWHSKEGDPLVFATVGSNRVTLYECHSQGEIRLLQSYVDADADENFYTCAWTYDSNTSHPLLAVAGSRGIIRIINPITMQCIKHYVGHGNAINELKFHPRDPNLLLSVSKDHALRLWNIQTDTLVAIFGGVEGHRDEVLSADYDLLGEKIMSCGMDHSLKLWRINSKRMMNAIKESYDYNPNKTNRPFISQKIHFPDFSTRDIHRNYVDCVRWLGDLILSKSCENAIVCWKPGKMEDDIDKIKPSESNVTILGRFDYSQCDIWYMRFSMDFWQKMLALGNQVGKLYVWDLEVEDPHKAKCTTLTHHKCGAAIRQTSFSRDSSILIAVCDDASIWRWDRLR
Vector:pET28-MHL
Growth
Medium:
Antibiotics:
Procedure:LEX Bubbling. For native protein: Human EED (76-441) covering the WD40 repeats was subcloned into pET28-MHL, a modified pET28a vector. Escherichia coli BL21 (DE3) Codon Plus RIL (Stratagene) strain was used as the expression host and grown in lysogeny broth at 37°C until the OD600 reaches 2.0. Protein expression was then induced with 0.5 mM IPTG at 18°C overnight. The cells were harvested by centrifugation (5,000 rpm Beckman JLA-8.1000rotor 15 min) and flash frozen in liquid nitrogen and stored at -80 degree.
Purification
Buffers
Washing Buffer: 50 mM Tris-HCl pH 7.4, 350 mM NaCl, 5% glycerol, 1 mM imidazole - Elution Buffer: 50 mM Tis-HCl pH 7.4, 350 mM NaCl, 5% Glycerol, 250 mM imidazole - Dialysis Buffer: 50 mM Tris-HCl pH 7.4, 350 mM NaCl, 5% glycerol - Gel Filtration Buffer: 20 mM Tris-HCl pH 8.0, 350 mM NaCl, 5% glycerol, 1 mM DTT
Procedure
The N-terminal His-tagged fusion proteins were purified by affinity chromatography on Ni-NTA agarose resin (Qiagen). The histidine tag was subsequently cleaved by incubating with TEV protease at a 1:20 TEV-to-protein weight ratio at 4°C overnight in dialysis buffer to remove imidazole. The cleaved protein was purified by passing it through another Ni-NTA resin. The flow-through was collected and purified further by size exclusion chromatography (Superdex 200; GE Healthcare). The proteins were concentrated to 15-20 mg/mL in a buffer containing 20 mM Tris-HCl, pH 8, 350 mM NaCl, 5% glycerol and 1 mM DTT.
Extraction
Buffers
50 mM Tris-HCl pH 7.4, 350 mM NaCl, 5% Glycerol
Procedure
2L native cell pellet was resuspended in a total volume of 200 ml extraction buffer with1mM PMSF/Benzamidine freshly added and the cells disrupted by sonication for 10 mins at 5" on 7" off duty cycle at 120W output power.
Concentration:Concentration used for crystallization : native protein: 15-20 mg/mL
Ligand
UNC4859MassSpec:native protein: 42298.4 g/mol, expected 42296.2 g/mol.
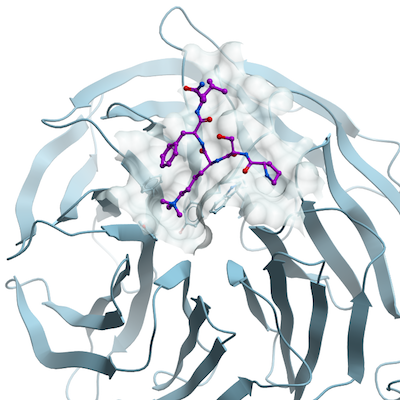
Crystallization:15 mg/ml EED (76-441) was mixed with UNC 4859 by adding 5-fold molar excess of UNC 4859 to the protein and incubating the mixture for 1 hour at 4°C. Protein crystallization was carried out by mixing equal volumes of protein and precipitant in a vapor diffusion setup, followed by incubation at 18°C. Crystals of EED grew in a buffer containing 20% PEG 3350, 0.1 M ammonium sulfate, 0.1 M Bis Tris pH 5.5 and were further optimized by micro-seeding. Prior to flash-freezing in liquid nitrogen, harvested crystals were cryo-protected by soaking in a buffer containing 30% PEG 3350, 0.1 M ammonium sulfate and 0.1 M Bis Tris pH 5.5.
NMR Spectroscopy:
Data Collection:
Data Processing: